Mass Spectrometry Part 1
440 likes | 2.73k Vues
Mass Spectrometry Part 1. Lecture Supplement: Take one handout from the stage. Determine structure of unknown substance. Verify purity/identity of known substance. Spectroscopy. Why bother with spectroscopy?. Mass spectrometry (MS)* molecular formula. Infrared spectroscopy (IR)

Mass Spectrometry Part 1
E N D
Presentation Transcript
Mass Spectrometry Part 1 Lecture Supplement: Take one handout from the stage
Determine structure of unknown substance • Verify purity/identity of known substance Spectroscopy Why bother with spectroscopy?
Mass spectrometry (MS)* molecular formula Infrared spectroscopy (IR) functional groups Nuclear magnetic resonance (NMR) C/H molecular skeleton X-ray crystallography* spatial position of atoms SpectroscopyWhat methods are commonly used? *Not rigorously a type of spectroscopy
Spectroscopy • Example: unidentified white powder • MS: C10H15N • IR: benzene ring, secondary amine (R2NH) • NMR: has CH2-CH-CH3 • X-ray: not necessary in this case
Fundamental operating principle Determine mass by manipulating flight path of an ion in a magnetic field Electron gun Magnet Ionization Measure ion sample m/z too small m/z too large mass-to-charge ratio m/z just right introduction (m/z) - + Accelerator plates Detector Detector quiet Detector fires Detector quiet Mass SpectrometryThe Mass Spectrometer Ionization: X + e- X+. + 2 e-
Isotopes Isotopes: atoms with same number of protons and same number of electrons but different numbers of neutrons • Aston mass spectrum of neon (1919) • Ne empirical atomic weight = 20.2 amu • Ne mass spectrum: predict single peak at m/z = 20.2 Resultsm/zrelative intensity 20.2 no peak 20.0 90% 22.0 10% • Conclusions • Neon is a mixture of isotopes • Weighted average: (90% x 20.0 amu) + (10.0% x 22.0 amu) = 20.2 amu • Nobel Prize in Chemistry 1922 to Aston for discovery of stable element isotopes
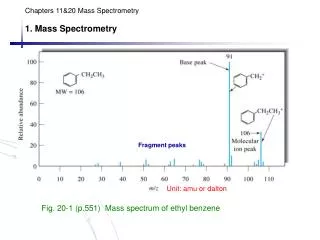
m/z = (1 x 12) + (4 x 1) = 16 C H Base peak: most abundant ion The Mass Spectrum Example: methane CH4 + e- CH4+. + 2 e- Relative ion abundance (%) mass-to-charge ratio (m/z)
m/z (amu)Relative abundance (%) < 0.5 17 1.1 16 100.0 85.0 9.2 3.0 12 1.0 M - 2H M - 3H M - 4H The Mass Spectrum Alternate data presentation... M+2 14C1H4 or 12C3H1H3 or... M+1 13C1H4 or 12C2H1H3 M 12C1H4 M - H Molecular ion (M): intact ion of substance being analyzed Fragment ion: formed by cleavage of one or more bonds on molecular ions
The Mass SpectrumOrigin of Relative Ion Abundances This table will be provided on an exam. Do not memorize it.
The Mass SpectrumRelative Intensity of Molecular Ion Peaks Imagine a sample containing 10,000 methane molecules... Molecule# in samplem/zRelative abundance 12C1H4 9889 12 + (4 x 1) = 16 100% 110 13 + (4 x 1) = 17 (110/9889) x 100% = 1.1%* 13C1H4 ~1 14 + (4 x 1) = 18 (1/9889) x 100% = < 0.1%* 14C1H4 *Contributions from ions with 2H are ignored because of its very small natural abundance CH4 mass spectrum m/z = 16 (M; 100%), m/z = 17 (M+1; 1.1%), m/z = 18 (M+2; < 0.1%)
When relative contribution of M = 100% then relative abundance of M+1/1.1% gives the approximate number of carbon atoms in the molecular formula Formula from Mass SpectrumM+1 Contributors • Comparing many mass spectra reveals M+1 intensity ~1.1% per C in formula • Examples: C2H6 M = 100%; M+1 = ~2.2% • C6H6 M = 100%; M+1 = ~6.6% • Working backwards gives a useful observation... • Other M+1 contributors • 15N (0.37%) and 33S (0.76%) should be considered • 2H (0.015%) and 17O (0.037%) can be ignored
Formula from Mass SpectrumM+2 Contributors Anything useful from intensity of M+2? IsotopesNatural abundancesIntensity M : M+2 32S : 34S 95.0 : 4.2 100 : 4.4 35Cl : 37Cl 75.8 : 24.2 100 : 31.9 79Br : 81Br 50.7 : 49.3 100 : 97.2 Conclusion: Mass spectra of molecules with S, Cl, or Br have significant M+2 peaks
C H Cl M: 36 + 7 + 35 = 78 Relative abundance (%) 78 80 m/z Formula from Mass SpectrumM+2 Contributors C3H7Cl M+2: 36 + 7 + 37 = 80 M:M+2 abundance ~3:1
C H Br M: 36 + 7 + 79 = 122 122 124 Formula from Mass SpectrumM+2 Contributors C3H7Br Relative abundance (%) M+2: 36 + 7 + 81 = 124 M:M+2 abundance ~1:1 m/z
Identifying the Molecular Ions • Which peaks are molecular ions? • Highest m/z not always M • M+1 has m/z one more than m/z of M C7H7Br M: m/z = 170
Formula from Mass Spectrum Summary of Information from Mass Spectrum M: Reveals mass of molecule composed of lowest mass isotopes M+1: Intensity of M+1 / 1.1% = number of carbons M+2: Intensity reveals presence of sulfur, chlorine, and bromine Next lecture: procedure for deriving formula from mass spectrum
Mass Spectrometry Part 2 Lecture Supplement: Take one handout from the stage
Summary of Part 1 • Spectroscopy: Study of the interaction of photons and matter • Useful to determine molecular structure • Types: MS*, IR, NMR, x-ray crystallography* *not really spectroscopy • MS fundamental principle: Manipulate flight path of ion in magnetic field • Charge (z), magnetic field strength are known; ion mass (m) is determined • Isotopes: Natural abundance of isotopes controls relative abundance of ions • Molecular ion (M, M+1, M+2, etc.): Intact ion of substance being analyzed • m/z of M = molecular mass composed of lowest mass isotopes 1H, 12C, 35Cl, etc. • Relative abundance of M+1/1.1% gives approximate number of carbons • M+2 reveals presence of sulfur, chlorine, or bromine • Fragment ion: From decomposition of molecular ion before reaching detector • Analysis of fragmentation patterns not important for Chem 14C
? C3H7Cl Mass Spectrum Formula Structure How do we derive structure from the mass spectrum? • Not trivial to do this directly • Structure comes from formula; formula comes from mass spectrum
M: m/z = 78 C2H6O3 C3H7Cl C5H4N C6H6 etc. M Mass Spectrum Formula Structure • How do we derive formula from the mass spectrum? • m/z and relative intensities of M, M+1, and M+2 • A few useful rules to narrow the choices
Formula: NH3 N2H4 C7H5N3O6 C8H10N4O2 m/z (M): 17 32 227 194 } The Nitrogen Rule How Many Nitrogen Atoms? Consider these molecules: NH3 H2NNH2 • Conclusion • When m/z (M) = even, number of N in formula is even • When m/z (M) = odd, number of N in formula is odd
m/z even odd nitrogen count even nitrogen count even nitrogen count even nitrogen count discarded How Many Nitrogen Atoms?A Nitrogen Rule Example Example: Formula choices from previous mass spectrum M: m/z = 78 C2H6O3 C3H7Cl C5H4N C6H6
C6H12 C6H10 H count = max - 2 H count = max - 4 How Many Hydrogen Atoms? One pi bond Two pi bonds C6H14 max H for 6 C Conclusion: Each pi bond reduces max hydrogen count by two
C6H12 C6H14 max H for 6 C C6H10 H count = max - 2 H count = max - 4 How Many Hydrogen Atoms? One ring Two rings Conclusion: Each ring reduces max hydrogen count by two
C6H15N C6H14 max H for 6 C C6H16N2 H count = max + 1 H count = max + 2 The Hydrogen Rule How Many Hydrogen Atoms? One nitrogen Two nitrogens Conclusion: • Each nitrogen increases max H count by one • For C carbons and N nitrogens, max number of H = 2C + N + 2
Mass Spectrum Formula • Procedure • Chem 14C atoms: H C N O F S Cl Br I • M = molecular weight (lowest mass isotopes) • M+1: gives carbon count • M+2: presence of S, Cl, or Br • No mass spec indicator for F, I Assume absent unless otherwise specified • Accounts for all atoms except O, N, and H • MW - mass due to C, S, Cl, Br, F, and I = mass due to O, N, and H • Systematically vary O and N to get formula candidates • Trim candidate list with nitrogen rule and hydrogen rule
Given information Mass Spectrum FormulaExample #1 m/zMolecular ionRelative abundanceConclusions 102 M 100% Mass (lowest isotopes) = 102 Even number of nitrogens 103 M+1 6.9% 6.9 / 1.1 = 6.3 Six carbons* 104 M+2 0.38% < 4% so no S, Cl, or Br Oxygen? *Rounding: 6.00 to 6.33 = 6; 6.34 to 6.66 = 6 or 7; 6.67 to 7.00 = 7
Mass Spectrum FormulaExample #1 Mass (M) - mass (C, S, Cl, Br, F, and I) = mass (N, O, and H) 102 - C6 = 102 - (6 x 12) = 30 amu for N, O, and H OxygensNitrogens30 - O - N = HFormulaNotes 0 0 30 - 0 - 0 = 30 C6H30 Violates hydrogen rule 1 0 30 - 16 - 0 = 14 C6H14O Reasonable 2 0 30 - 32 - 0 = -2 C6H-2O2 Not possible 0 30 - 0 - 28 = 2 C6H2N2 Reasonable 2* *Nitrogen rule! • Other data (functional groups from IR, NMR integration, etc.) further trims the list
m/zMolecular ionRelative abundanceConclusions 157 M 100% 158 M+1 9.39% 159 M+2 34% Mass Spectrum FormulaExample #2 Mass (lowest isotopes) = 157 Odd number of nitrogens 9.39 / 1.1 = 8.5 Eight or nine carbons One Cl; no S or Br
1* *Nitrogen rule! Mass Spectrum FormulaExample #2 Try eight carbons: M - C8 - Cl = 157 - (8 x 12) - 35 = 26 amu for O, N, and H OxygensNitrogens26 - O - N = HFormulaNotes 0 26 - 0 - 14 = 12 C8H12ClN Reasonable Not enough amu available for one oxygen/one nitrogen or no oxygen/three nitrogens
1* *Nitrogen rule! Mass Spectrum FormulaExample #2 Try nine carbons: M - C9 - Cl = 157 - (9 x 12) - 35 = 14 amu for O, N, and H OxygensNitrogens14 - O - N = HFormulaNotes 0 14 - 0 - 14 = 0 C9ClN Reasonable Not enough amu available for any other combination.
Formula Structure • What does the formula reveal about molecular structure? • Functional groups • Absent atoms may eliminate some functional groups • Example: C7H9N has no oxygen-containing functional groups • Pi bonds and rings • Recall from previous: one pi bond or one ring reduces max H count by two • Each two H less than max H count = double bond equivalent (DBE) • If formula has less than full H count, molecule must contain one pi bond or ring
hydrogens and halogens nitrogens H N DBE = C - + + 1 2 2 carbons Example C8H10ClN Formula StructureCalculating DBE DBE may be calculated from molecular formula: • One DBE = one ring or one pi bond • Two DBE = two pi bonds, two rings, or one of each • Four DBE = possible benzene ring DBE = C - (H/2) + (N/2) + 1 = 8 - [(10+1)/2] + (1/2) + 1 Four pi bonds and/or ring Possible benzene ring = 4
Formula StructureCommon Math Errors • Small math errors can have devastating effects! • No calculators on exams • Avoid these common spectroscopy problem math errors: • Divide by 1.1 divide by 1.0 • DBE cannot be a fraction • DBE cannot be negative Next lecture: Infrared spectroscopy part 1