Exploring Light Waves and the Quantum Model of Electrons in Atoms
410 likes | 439 Vues
Dive into the properties of light waves and the quantum model governing electrons in atoms. Explore characteristics like amplitude, wavelength, frequency, and speed of light. Understand the Photoelectric Effect, emission spectra, and the principles of the Quantum Model. Discover how quantum numbers define atomic orbitals and electron properties, and learn about the arrangement of electrons in atoms.

Exploring Light Waves and the Quantum Model of Electrons in Atoms
E N D
Presentation Transcript
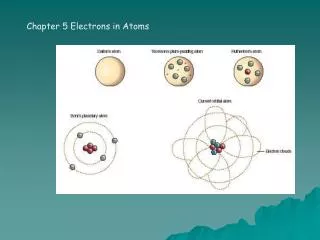
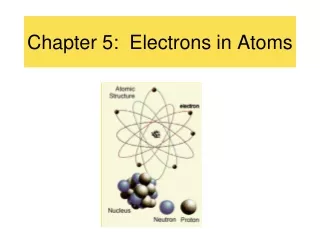
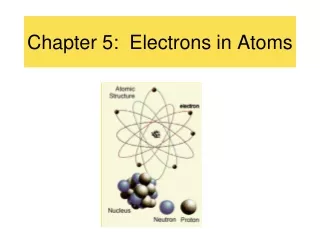
Chapter 4 Electrons In Atoms
Properties of Light • ___________________: a form of energy that exhibits wavelike behavior as it travels through space. (ex. Visible light, x-rays, UV, IR, radio) • ____________________: all forms of electromagnetic radiation together make up the spectrum. 4-2
Characteristics of Waves • There are 4 main characteristics of waves: • 1) ___________: The height of the wave measured from it’s origin to it’s peak. When you increase the intensity, or brightness of light, you are increasing it’s amplitude. 4-4
Characteristics of Waves • 2)________________: the distance between corresponding points on adjacent waves. Wavelength is designated by the Greek symbol __________. Wavelength = ___ 4-6
Characteristics of Waves • 3) ___________: the number of waves that pass a given point in a specific time. Measured in cycles per second (cycle/second, or s-1) The SI unit for this is Hertz. 1.0 Hz = 1.0 s-1 Frequency is designated by the Greek symbol ___. Frequency = ____ 4-8
Characteristics of Waves • 4)_________: the speed of light is constant. It is rounded to _________m/s. The speed of light is represented by the letter ____. ___ = λυ 4-10
The Photoelectric Effect • Refers to the emission of _________ from a metal when light shines on the metal. • __________: the minimum amount of energy that can be lost or gained by an atom • ____________, a German physicist studied quanta of light and found: 4-11
The Photoelectric Effect E(energy) = _____ Where h is Plank’s Constant and has a value of 6.6262 x 10-34 Js 4-12
The Photoelectric Effect Einstein expanded upon this to propose that light has a ______________, acting as a _______ under some circumstances and a ____________ under others. __________: a particle of electromagnetic radiation having zero mass and carrying a quantum of energy. 4-13
Line Emission Spectrum ______________: the lowest energy state of an atom _____________: a state in which an atom has a higher potential energy than it has in it’s ground state. 4-14
Line Emission Spectrum _____________________: a graph that indicates the degree to which a substance emits radiant energy with respect to ______________. ______________________: the emission of a continuous range of frequencies of electromagnetic radiation. 4-15
H Emission Spectrum _____________: a Danish physicist who proposed a hydrogen-atom model that linked the atom’s electron to photon emission. (electrons circle the nucleus in “_______”) 4-16
H Emission Spectrum Emission Line Spectrum: a graph that indicates the degree to which a substance emits radiant energy with respect to wavelenth. Continuous Spectrum: the emission of a continuous range of frequencies of electromagnetic radiation. 4-17
H Emission Spectrum Emission Line Spectrum: a graph that indicates the degree to which a substance emits radiant energy with respect to wavelenth. Continuous Spectrum: the emission of a continuous range of frequencies of electromagnetic radiation. 4-18
The Quantum Model ____________________________: states that it is impossible to determine simultaneously both the ___________ and _______________ of an electron or any other particle. ___________________: describes mathematically the wave properties of electrons and other very small particles. 4-19
The Quantum Model ______________________: specify the properties of atomic orbitals and the properties of electrons in orbitals. Quantum numbers were developed based on the ____________________, developed by Austrian physicist Erwin Shrödenger. 4-20
The Quantum Model __________________________: indicates the main energy level occupied by the electron. Values are positive integers only (1, 2, 3, 4 with 1 being the lowest energy level closest to the nucleus) _______________________: indicates the shape of the orbital. Values are 0, 1…n-1) 4-21
The Quantum Model l = 0 = sl = 1 = p l = 2 = d l = 3 = f l = 0 = S l = 1 = p l = 2 = d l = 3 = f 4-21
The Quantum Model Magnetic Quantum Number (m): indicates the orientation of an orbital around the nucleus. Values, including zero, are –l to +l l = 0 = s orbital has only one orientation (sphere) l = 1 = p has three orientations l = 2 = d has five orientations l = 3 = f has seven orientations 4-23
The Quantum Model ________________________: indicates the two fundamental spin states of an electron in an orbital. Values are +1/2 or -1/2. ______________________: the arrangement of electrons in an atom. 4-27
Orbital Filling Diagrams There are ____________, named after the scientists that discovered them, that govern the filling of these orbitals with electrons… • The ________________: an electrons occupies the lowest energy orbital that can receive it. 4-28
Orbital Filling Diagrams 2) The ________________________: no two electrons in the atom can have the same set of four quantum numbers. 3) _______________: Electrons occupy equal energy orbitals so that a maximum numbered of unpaired electrons results, and all e- in singly occupied orbitals must have the same spin. 4-29
Orbital Filling Diagrams When using this form, each electron is designated as an _______ ________ pointing up or down to show opposite spins. Each orbital is designated with a labeled line: ____ or __ __ __ 1s 2p ____________lines show multiple orbitals (1 for s, 3 for p, 5 for d) 4-30
Orbital Filling Diagrams Orbitals fill going _____________ each period (_____) on the periodic table, from the lowest energy level up. (Aufbau). Don’t forget, when they pair, they have ____________ (Pauli), but they won’t pair until each available orbital has an unpaired electron in it first (Hund) 4-31
Orbital Filling Diagrams 4-32 4-31
Orbital Filling Diagrams _____________, with one electron, would have an orbital filling diagram of: 1s __________, with 2 electrons, would be: 1s Now your at the end of the first period, start again in the 2nd period with 2s… 4-33
Orbital Filling Diagrams Lithium: 1s 2s Be: 1s 2s B: _ _ 1s 2s 2p
Orbital Filling Diagrams Which of these would be correct for oxygen (with 8 e-): O: ? 1s 2s 2p OR 1s 2s 2p ____________ is correct, the _________ example violates the Pauli Exclusion Principle. 4-35
Practice Write the orbital filling notation for the following elements: • Be:_________________________ • F:__________________________ • Ar:_________________________ • Cu:_________________________ 4-36
Electron Configurations Now your ready to write electron configurations. These are simply the orbital diagrams written out with superscripts: Lithium: 1s 2s would be written out as 1s2 2s1 Be: 1s 2s would be written out as 1s2 2s2 B: _ _ 1s 2s 2p would be written out as 1s22s22p1 4-37
Practice Write the electron configuration for the following elements: • Mg:________________________ • N:_________________________ • Cr:________________________ • Cl:________________________ 4-38
Electron Configurations ___________________: refers to an outer main energy level occupied by eight e- • Once a __________ is complete at the end of a period, you can write subsequent configurations as having the _______ of the ___________ with the additional ___________ electrons. • Sodium Na would have a noble gas notation of: Ne3s1 4-39
Practice Write the noble gas notation for the following elements: • Na:_________________________ • Sb:_________________________ • Y:__________________________ • F:__________________________ 4-40
Ch. 4 The End 4-41