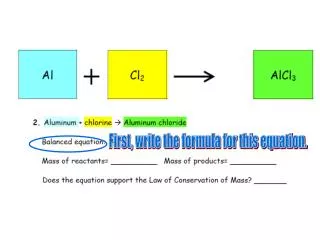
Understanding Metal and Anion Formulas: Cation Charges, Polyatomics, and Naming Conventions
E N D
Presentation Transcript
Write the formula Metal (cation)first (roman) indicates metal charge Anion next, use first on PT for “ides” use half sheet for polyatomics Cation charge becomes anion subscript and vice versa(“criss-cross” charges) Reduce as necessary by greatest common factor
Sodium Chloride Na+ Cl- NaCl
Magnesium fluoride Mg2+ F- MgF2
calcium hydroxide Ca2+ OH- Ca(OH)2
Name a formula Metal (cation)first, just the name no prefix, no suffix IFF!!! PT shows more than one charge for the metal, Use (roman) to indicate metal charge Cation charge = (anion charge)X(anion subscript)/(cation subscript) Anion name next
Fe3(PO4)2 Iron Iron ( ) Iron ( ) Phosphate (3)x(2)/3=2 Iron (II) Phosphate +2 +3 Fe There’s two oxidation states, so we’ll need roman numerals. Anion subscript is 2 Anion charge is 3- Cation subscript is 3 Roman = 2
Parenthesis • Use around polyatomics when there’s more than one in the molecule!