Advanced Nasal Systems for Treating Type 1 Diabetes
This study explores novel nasal delivery systems for treating Type 1 diabetes, focusing on system description, efficacy evidence, and developmental stages. The system leverages the nasal cavity's anatomy for improved insulin delivery, with potential for enhanced patient compliance compared to traditional routes.

Advanced Nasal Systems for Treating Type 1 Diabetes
E N D
Presentation Transcript
Novel delivery systems for treating Type 1 diabetes Nasal Systems Han Yi Chua Gek Huey
Outline • Rationale • System description • Evidence for efficacy • Stage of development
Rationale • Improving patient compliance compared with parenteral routes • Large absorptive surface area and the high vascularity of the nasal mucosa • Avoiding hepato-gastrointestinal first-pass metabolism • Similar plasma pharmacokinetics (PK) profile to intravenous injection • Mimicing pulsatile secretion pattern of insulin
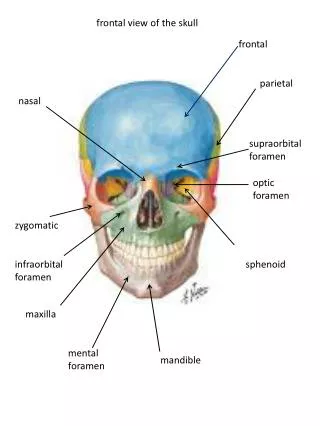
Nasal cavity anatomy Increased absorption area turbinates Epithelia monolayer High permeability & rapid absorption
Structure of nasal mucosa • Mucociliary clearance – cilia & mucus • Insoluble particles removed before being absorbed • Gases/solution penetrate further into respiratory tract
Barriers to intranasal insulin absorption • Removal of deposited insulin by mucociliary clearance • Difficulty of penetrating mucus layer and epithelial membrane • Enzymatic degradation
Removal of deposited insulin by mucociliary clearance • Mucociliary mechanism t1/2 of clearance is only 15-30min decreases time available for drugs to be systemically absorbed • Size of particles suitable for nasal delivery particle aerodynamic diameter: 1-10µm
Penetration of mucus layer and epithelial membrane • Mucus layer Drugs may interact with mucus glycoproteins • Epithelial membrane Lipophilic compounds via transcellular route Hydrophilic compounds (MW< 1000 daltons) via paracellular route
Enzymatic degradation • Nasal cavity is still a significant barrier although intranasal route avoids first pass metabolism
System modification • Absorption enhancers or promoters most frequently used approach • Modification of the structure of insulin
Formulation types in current research • Spray • Drop • Powder
1922 1931 1932 1935/6 • Major[123,124] • Uses glycols • Normal rabbits, dogs and diabetic patients • Marked decreases in blood glucose conc. • high dosage: 100IU nasal • Poor reproducibility of hypoglycaemic control Woodyatt[23] Fail to generate positive results Horwitz[121] Failed to demonstrate insulin absorption thru nasal route • Collens[122] • Uses saponin (natural glycoside surfactant) • 100IU nasal 10IU subcutaneous injection • Causes nasal congestion & • symptoms of rhinitis Evidence of efficacy (I)
1958 • Hankiss[125] • Nasal tampons (increased contact time) • Healthy volunteers and diabetic patients • 53% that of subcutaneous injection • No absorption promoter used • Unstable & variable • Efficacy calculated 60 mins after administration (subcutaneous injection had not reached nadir) Evidence of efficacy (II)
Bile salts SGC SDC STDHF 1977 1983 1987 1988 1989 1994 Laureth-9 Surfactants LPC DDPC LPG Fatty acids Evidence of efficacy (III)
Bioadhesive Carbopol Starch microshperes Chitosan 1985 1988 1996 • Chitosan • Rel. Bioavailability (subcutaneous)= 15% • Rel. Bioavailability (intravenous) = 7% • Starch microspheres: • 30% to intravenous dose • Abs bioavailability = 4.5% • Starch microshperes with LPC: • Abs. bioavailability = 13% • Rel. bioavailability (intravenous)= 32% Evidence of efficacy (IV)
Requirements of enhancer • Rapid-acting • Transient & reversible modulation • Not absorbed systemically • Predictable & reproducible degree of absorption enhancement • Safe for chronic nasal administration • Open tight junction, disrupt membrane
Systemic insulin absorption after intranasal administration to humans
Modification of structure • Lys(B28)Pro(B29) insulin analogues [Merkus, 1996] • Powder formulation with cyclodextrin • Chemical modification with fatty acids [Asada, 1992] • Enhance lipophilicity • Pharmacological availability decreased
Stage of development • Bentley Pharmaceuticals (BNT) 02 Feb 2004 • Phase I clinical trial using proprietary intranasal insulin formulation in human volunteers http://www.biospace.com/ccis/news_story.cfm?StoryID=15018420&full=1 Successful Phase I Study Of Intranasal Insulin In Human Volunteers End
Bioadhesive • Interacts with mucosal layer Back
Chitosan • Enhance via paracellular route due to interaction of negatively charged epithellial cells resulting in structural changes in tight junction associated protein • Increase membrane permeability • Bioavailability abt 15% relative to subcutaneous; 7% relative to intravenous • Does not cause change in saccharin clearance times • No adverse effect in nasal cilia beat frequency • Non-damaging to biomembranes • Reversible reduction in nucociliary transport rate (MTR) Back
Bile Salts • Amphiphilic molecules • Possess distinct non-polar and polar regions • critical micellar concentration • Arrest cilia beat frequency • Damage nasal mucosa Back
Surfactants • Lower surface and interfacial tension of water • Promote association into micelles • Modify properties of biological membranes • Alter their permeability and transport characteristics Back
Fatty acids • Organic compounds which are principal component of most lipids and consist mainly of straight chains of an even number of carbon atoms Back