Physical Science
240 likes | 251 Vues
Explore the world of compounds through ionic and covalent bonding, chemical formulas, structures, and properties. Learn about ionic compounds, covalent bonds, and naming conventions in Physical Science Chapter 6.

Physical Science
E N D
Presentation Transcript
Physical Science Chapter 6: Compounds Ionic and Covalent Compounds
Physical Science:Chapter 6, Compounds Chemical Bonds • A _____________is a substance made of two or more elements chemically combined (bonded together). • The forces that hold atoms or ions together are called _________________.
Physical Science:Chapter 6, Compounds Chemical Formulas • A compound can be represented by a __________________, like CO2 • The chemical formula tells you ______________________________and the ______________________.
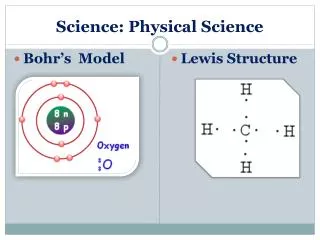
Physical Science:Chapter 6, Compounds Chemical Structures • A __________________is aMODEL that shows you the elements in a compound, the ratios of the elements AND ______________________________
Physical Science:Chapter 6, Compounds Chemical Structures & Properties • The structure of a compound determines the _____________________________________ of the compound. color viscosity flammability
Physical Science:Chapter 6, Compounds Chemical Structures & Bonding • Usually atoms combine to form compounds so that each atom will have _________________________ ______________________________________________________________
Physical Science:Chapter 6, Compounds Chemical Bonding There are two basic types of chemical bonding • ______________________ • ______________________ We will also briefly cover • __________________________ • ____________________
Physical Science:Chapter 6, Compounds Chemical Bonding: Ionic Bonding • Ions are _________________. • Ions are formed when atoms transfer _______________. • __________are negative ions and _____________ are positive ions.
Physical Science:Chapter 6, Compounds Chemical Bonding: Ionic Bonding • ___________________are formed when cations andanionsare attracted to each other. • Since__________formcations and_________formanions,many ionic compounds are made of ________ions and __________ions, like Na+ &Cl- to form NaCl.
Physical Science:Chapter 6, Compounds Properties of Ionic Compounds • ________________________________ Ionic bonds are fairly strong so that ionic compounds • ________________________________ • ________________________________ • ________________________________
Physical Science:Chapter 6, Compounds Chemical Bonding: Covalent Bonds • _____________atoms (except the Noble Gases) do NOT have“enough”________ electrons (short of an octet) • If non-metals are surrounded by other non-metal elements, they can’t transfer electrons, so they will ________________electrons in _______________________.
Physical Science:Chapter 6, Compounds Chemical Bonding: Covalent Bonds • Sometimes non-metals atoms only need to share a pair of electrons. One shared pair = _________ covalent bond. • ____shared pairs = a __________bond • ____shared pairs = a __________bond • Covalently bonded units are known as molecules.
Physical Science:Chapter 6, Compounds Chemical Bonding: Covalent Bonds • When two atoms of the same element form covalent bonds by sharing electrons, they share electrons __________________, so the bond is a_____________________covalent bond. • When there is _______________sharing of electrons, it is called a __________ covalent bond.
Physical Science:Chapter 6, Compounds Chemical Bonding: Covalent Networks • _______________________________are very strong covalent bonding situations over _______atoms instead of just a pair of atoms. • Non-metals in covalent networks form some of the ____________substances known. • Examples of Carbon Networks: 1____________________2__________________ 3____________________
Metallic Bonds • ___________that are in elemental form or bonded together have a special kind of arrangement of nuclei surrounded by valence electrons that are ___________________. • This is often referred to as an “_______________” model or “_____________________” • The electrons are said to be “______________”. • It is believed that this special arrangement gives metals the properties that they possess, such as: • __________________________________ • ____________________ • ____________________
Physical Science Chapter 6: Compounds Naming Ionic and Covalent Compounds
Physical Science:Chapter 6, Compounds Ionic Compound Formulas • Ionic compound formulasshow the ratio of elements in an ionic compound. • For example: • The chemical formula BaCl2shows that there are two chloride ions for each barium ion in a barium chloride crystal.
Physical Science:Chapter 6, Compounds Ionic Compound Formulas • A single crystal of table salt has billions of ions in a crystal network so its formula could be: Na100,000,000,000 Cl100,000,000,000 • We use the lowest whole number ratio for the ionic formula, so the ionic compound formula isNa1Cl1 (NaCl) • This is known as an empirical formula.
Physical Science:Chapter 6, Compounds Ionic Compound Formulas • Ionic compound formulas have to “balance.” • They must have the same number of positives (+’s) as negatives (–’s) in the total formula (total equal to zero) • Examples: Na1+Cl1- (1+ and 1-) = 0 Ba2+S2- (2+ and 2-) = 0
Physical Science:Chapter 6, Compounds Covalent Molecule Formulas: • Molecular formulas are NOT just ratios like the empirical formulas for ionic compounds. • Molecular formulas state EXACTLY how many atoms of each element are in the molecule. Example: Glucose (C6H12O6)has: 6 carbon, 12 hydrogen & 6 oxygen atoms
Physical Science:Chapter 6, Compounds Covalent Molecule Formulas: • There are millions of compounds made with covalent bonds. • In Physical Science we will only deal with the formulas and names of the simplest types. • In this Chapter we will learn formulas and names for binarymolecules
Physical Science:Chapter 6, Compounds Covalent Molecule Names: • These compounds will contain two nonmetals • For binary covalent molecules’ names: a). List the elements in the compound b). Say how many of each element are in the compound.
Physical Science:Chapter 6, Compounds Covalent Molecule Names: • To say the number of atoms, chemists use the following numbering system: mono = 1 hexa = 6 di = 2 hepta = 7 tri = 3 octa = 8 tetra = 4 nona = 9 penta = 5 deca = 10 *Note: “mono” is not usually used for the first element in a covalent compound.