Quiz 2/9/05
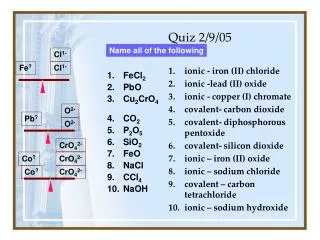
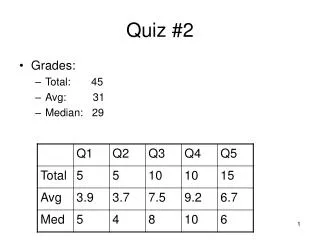
iron (III) bromide calcium oxide nickel (II) chromate carbon disulfide carbon tetrabromide calcium hydroxide diphosphorous pentoxide. FeBr 3 CaO NiCrO 4 CS 2 CBr 4 Ca(OH) 2 P 2 O 5. Quiz 2/9/05. Br 1-. Br 1-. Fe ?. Br 1-. Ni ?. CrO 4 2-. 14 points each.

Quiz 2/9/05
E N D
Presentation Transcript
iron (III) bromide calcium oxide nickel (II) chromate carbon disulfide carbon tetrabromide calcium hydroxide diphosphorous pentoxide FeBr3 CaO NiCrO4 CS2 CBr4 Ca(OH)2 P2O5 Quiz 2/9/05 Br1- Br1- Fe? Br1- Ni? CrO42- 14 points each
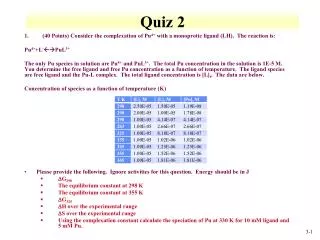
Electronegativity: the relative tendency of an atom to attract electrons to itself when participating in a chemical bond. • Most bonds have some ionic character. • If more than 50% of the time one ion has possession of the electron, the compound is considered ionic. • This means that if the difference in electronegativity is greater than 1.67, the compound is considered ionic. • The more ionic a bond, the more easily a solution of the compound will conduct electricity. • Electricity is defined as the flow of electrons. • Do pre-lab for lab 21
Ca(OH)2 Fe2O3 MnO2 iron I(I) oxide CO carbon dioxide dinitrogen trioxide Li2S AsCl3 Ca3N2 potassium oxide lead (II) oxide GeCl4 SiC OF2 CaC2 iron (III) sulfide magnesium chloride zinc oxide Sr(CN)2 SO3 iron (II) sulfide iron (II) sulfite iron (II) sulfate silver oxide PCl3 NH4Cl (NH4)2SO4 • calcium hydroxide • iron (III) oxide • manganese (IV) oxide • FeO • carbon monoxide • CO2 • N2O3 • lithium sulfide • arsenic trichloride • calcium nitride • K2O • PbO • germanium tetra chloride • silicon monocarbide • oxygen difluoride • BaBr2 • SF6 • BrF3 • GaF3 • IF5 • Pb(NO3)2 • Cr(OH)3 • FeSO4 • ZnSO4 • MnSO4 • Na2CrO4 • PbCrO4
CaC2 iron (III) sulfide magnesium chloride zinc oxide Sr(CN)2 SO3 iron (II) sulfide iron (II) sulfite iron (II) sulfate silver oxide PCl3 NH4Cl (NH4)2SO4 • BaBr2 • SF6 • BrF3 • GaF3 • IF5 • Pb(NO3)2 • Cr(OH)3 • FeSO4 • ZnSO4 • MnSO4 • Na2CrO4 • PbCrO4 • barium bromide • sulfur hexafluoride • bromine tri fluoride • gallium trifluoride • iodide pentafluoride • lead (II) nitrate • chromium (III) hydroxide • iron (II) sulfate • zinc sulfate • manganese (II) sulfate • sodium chromate • lead (II) chromate
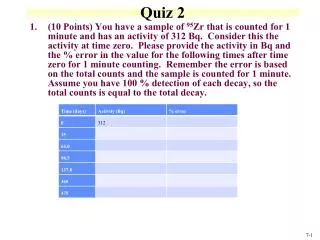
CaC2 iron (III) sulfide magnesium chloride zinc oxide Sr(CN)2 SO3 iron (II) sulfide iron (II) sulfite iron (II) sulfate silver oxide PCl3 NH4Cl (NH4)2SO4 • calcium carbide • Fe2S3 • MgCl2 • ZnO • strontium cyanide • sulfur trioxide • FeS • FeSO3 • FeSO4 • Ag2O • phosphorous trichloride • ammonium sulfate Lab 21: Conductivity of substances
Naming Organic Compounds meth = 1 eth = 2 prop = 3 but = 4 pent = 5 hex = 6 hept = 7 oct = 8 non = 9 … • Organic compounds have one or more carbons, surrounded with hydrogens. • They may have double or triple bonds, and may include oxygen, or other atoms. • They are named by counting the “carbon backbone” and applying a prefix • “Functional Groups” provide the rest of the name.
Naming Organic Compounds meth = 1 eth = 2 prop = 3 but = 4 pent = 5 hex = 6 hept = 7 oct = 8 non = 9 … • Butane = 4 carbons • CH3CH2CH2CH3 • Heptane = 7 carbons • CH3CH2CH2CH2CH2CH2CH3 • Octane = 8 carbons • CH3CH2CH2CH2CH2CH2CH2CH3
13 c page 178 6 hydrogen atoms 3 carbon atoms
13 f page 178 6 carbon atoms 12 hydrogen atoms http://jchemed.chem.wisc.edu/JCESoft/CCA/CCA5/MAIN/1ORGANIC/ORG02/TRAM02/E/NOMOVIE/MISC.HTM
Molecular and Empirical Formulas. • Molecular: adj. Pertaining to, consisting of, caused by, or existing between molecules. • Empirical: adj. 1. Relying upon or derived from observation or experiment. 2. Guided by practical experience and not theory, especially in medicine. • Formula: n 1. an established form of words for use in a ceremony or procedure. 3. Chemistry: a. a symbolic representation of the composition or of the composition and structure of a chemical compound. b. The chemical compound so represented. c. A prescription in exact proportion: recipe.
C6H12O6 H2O2 Hg2I2 C6H14 CH2O HO HgI C2H7 Molecular and Empirical Formulas
Coefficients: How many sets of a particular compound/element • 2 C6H12
Coefficients: How many sets of a particular compound/element • 5 C6H12