Quantum Number and Electron Configurations
250 likes | 690 Vues
Quantum Number and Electron Configurations. The arrangement of electrons in an atom. Quantum Numbers. Scientists use quantum number to describe the probable region for finding an electron There are 4 quantum number. The Principle Quantum Number. Symbolized by n

Quantum Number and Electron Configurations
E N D
Presentation Transcript
Quantum Number and Electron Configurations The arrangement of electrons in an atom
Quantum Numbers • Scientists use quantum number to describe the probable region for finding an electron • There are 4 quantum number.
The Principle Quantum Number • Symbolized by n • Indicates the main energy level occupied the electron (the ring away from the nucleus) • Values of n are positive integers (1,2,3,4) • As nincreases, so does the distance from the nucleus as well as the electron’s energy.
Angular Momentum Quantum Number • Represented by l • Represents the shape or type of orbital that corresponds to a particular suborbital • l=n-1, n-2, n-3, up to n-n • So, if n = 3,l can equal2 ,1 and 0 • Each of these numbers is given a letter. We will only deal with the first four letters. • 0=s, 1=p, 2=d, 3=f
Magnetic Quantum Number • Represented by m • This a subset of l • Indicates the number and orientations of orbitals around the nucleus • It is equal to all of the integers from –l to +l • So, if l = 2, then m = -2, -1, 0, 1, 2
Spin Quantum Number • Symbolized by +1/2 and -1/2 or and • Indicates the orientation of the electrons’ magnetic field • Also known as spin
How many electrons can each level hold? • The formula used to calculate the number of electrons each level can hold is 2n2
So, those are the Quantum Numbers But what do they mean? The answer is in something called electron configurations
Aufbau Principle • How did you decide where to send the people at the Hotel Californium? • “Each electron occupies the lowest energy orbital available” • Lowest Energy Highest Energy s p d f 1 3 5 7
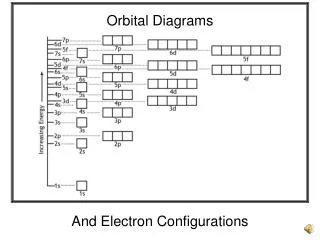
Orbital Filling Sequence and Energy Levels From Glencoe – Chemistry: Matter and Change
Pauli Exclusion Principle • “One person to a bed” • “A maximum of two electrons may occupy a single orbital sublevel, but only if the electrons have opposite spins”
Hund’s Rule • The Family Situation • “Single electrons with the same spin must occupy each equal-energy orbital before additional electrons with opposite spins can occupy the same orbital.”
What does that mean in English? • Much like the family at Hotel Californium, electrons will space each other out as much as possible before occupying spaces close to other electrons. • Here’s another way to think about it: How do strangers sit on a bus as it fills up?
With all this talk about electrons, here is a review question?Where can we find out how many electrons an atom has?
Now we know the rules and the number of electrons, what do we do with them?
Remember this from earlier? Example: Chlorine Electrons? 17 Electron configuration: 1s2 2s2 2p6 3s2 3p5 Lowest Energy to Highest Energy From Glencoe – Chemistry: Matter and Change
So, if you had the orbital filling sequence, can you fill in the electrons? • 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d, 7p Let’s take Cl again. Cl has 17 electrons. Orbital diagram 1s 2s 2p 3p 3s Writing it without the boxes: 1s2 2s2 2p6 3s2 3p5 Electron Configuration
How can I remember all of those numbers and letters? s p d f 1 2 2 3 3 3 4 4 4 4 5 5 5 5 5 6 6 6 6 6 6 7 7 7 7 7 7 7 The Diagonal Rule X X X 1s 2s 2p 3s 3p 4s 3d 4p 5s X X 4d 5p 6s 4f 5d 6p 7s 5f 6d 7p X X X