Alkenes, Alkynes, & Arenes
Alkenes, Alkynes, & Arenes. Some Terms. saturated : a molecule that only contains carbon-carbon single bonds (ex. CH 4 , CH 3 CH 2 CH 2 CH 3 , etc.) unsaturated : a molecule that contains C=C or C ≡ C bonds (ex. CH 2 = CH 2 , CH 3 CH 2 C ≡ CH 3 ). Alkene.

Alkenes, Alkynes, & Arenes
E N D
Presentation Transcript
Some Terms • saturated: a molecule that only contains carbon-carbon single bonds (ex. CH4, CH3CH2CH2CH3, etc.) • unsaturated: a molecule that contains C=C or C ≡ C bonds (ex. CH2 = CH2, CH3CH2C ≡ CH3)
Alkene • alkene: a hydrocarbon that contains a C = C bond
Alkynes • alkyne: a hydrocarbon that contains a C ≡ C bond
Naming Alkenes & Alkynes • rules are similar to those for alkanes • use “-ene” suffix for alkenes • use “-yne” suffix for alkynes
1. Name the Parent Chain • Find the longest carbon chain containing the double or triple bond • Name the parent compound by adding the ending “-ene” if a double bond, “-yne” if a triple bond CH3CH2CH2C ≡ CCH3 CH3CH2CH2 C=CHCH3 CH3CH2CH2 …hexyne …hexene
2. Number the Carbon Atoms in the Main Chain • Begin at the end nearer the multiple bond • If the multiple bond is an equal distance from both ends, begin numbering at the end nearer the first branch. CH3CH2CH2C ≡ CCH3 CH3CH2CH2 C=CHCH3 CH3CH2CH2 . …hexyne …hexene 6 5 4 3 2 1 6 5 4 2 1 3
3. Identify the Location of the Multiple Bond and Any Substituents. • Indicate the position of the multiple bond in the chain by giving the number of the first carbon to which it is attached. CH3CH2CH2C ≡ CCH3 CH3CH2CH2 C=CHCH3 CH3CH2CH2 . …hexyne …hexene 6 5 4 3 2 1 6 5 4 2 1 triple bond attached to 2nd carbon 3 propyl group attached to 3rd carbon double bond attached to 2nd carbon
4. Write the full name • Assign numbers to the branching substituents and list the substituents alphabetically. • Use commas to separate numbers • Use hyphens to separate words from numbers • Indicate the position of the multiple bond in the chain by giving the number of the first multiple-bonded carbon. • If more than one double bond is present, identify the position of each and use the appropriate name ending –diene, -triene, tetraene, etc.
6 5 4 3 2 1 6 5 4 CH3CH2CH2C ≡ CCH3 CH3CH2CH2 C=CHCH3 CH3CH2CH2 . 2-hexyne 3-propyl-2-hexene 2 1 3
Additional Example CH2CH3 H2C=CCH=CH2 2-ethyl-1,3-butadiene
Exceptions • CH2=CH2ethylene • CH3CH=CH2propylene • HC≡CH acetylene
Practice: Name CH3 CH2CH3 CH3CH2CH2C = CCH3 CH3 CH3CH2CH2CH=CHCHCH3 CH2=CHCH2CH2C=CH2 CH3 3,4-dimethyl-3-heptene 2-methyl-3-heptene 2-methyl-1,5-hexadiene
Practice: Draw 2-methyl-2-hexene CH2=CCHCH2CH2CH3 CH3 4,4-dimethyl-2-pentyne CH3 CH3C≡CCCH3 CH3 3-ethyl-2,2-dimethyl-3-hexene CH3 CH3 C C=CHCH2CH3 CH3 CH2CH3
Your Turn • Name CH3 CH3C=CCH3 CH3 2,3-dimethyl-2-butene • Draw 4-methyl-1-hexyne CH≡CCH2CHCH2CH3 CH3
Cycloalkenes • cycloalkene: a cyclic hydrocarbon that contains a double bond cyclohexene cyclopentene cyclobutene
Naming Cycloalkenes • The double-bond carbon atoms in the substituted cycloalkenes are numbered 1 and 2 so as to give the first substituent the lower number CH3 CH2CH3 CH3 4 4 5 3 CH3 3 2 1 2 1 4-methylcyclohexane 4-ethyl-3,5-dimethylcyclohexane
Cis-Trans Isomers • have same formula and connections between the atoms • have different three-dimensional structures because of the way that groups are attached to different sides of the double bond • occurs whenever each double-bonded carbon has two different substituent groups
Naming Cis-Trans Isomers same side of double bondopposite side of double bond
H CH2CH3 H CH3 C = C C = C H CH3 H CH2CH3 identical compound H H CH3 H C = C C = C CH3 CH2CH3 H CH2CH3 cis-2-pentene trans-2-pentene
Practice • Draw structures for both the cis and trans isomers of 2-hexene. CH3CH=CHCH2CH2CH3 CH3 CH2CH2CH3 CH3 H C=C C=C H H H CH2CH2CH3
Your Turn • Draw both cis and trans isomers of 3,4-dimethyl-3-hexene CH3 CH3 CH3CH2C = C CH2CH3 CH3CH2 CH2CH3 CH3CH2 CH3 C=C C=C CH3 CH3 CH3 CH2CH3 cis trans
Properties of Alkenes & Alkynes • nonpolar • insoluble in water • soluble in nonpolar organic solvents • less dense than wate • flammable • nontoxic • chemically reactive at the multiple bond
Reactions • addition • elimination • substitution • rearrangement
1. Addition Reactions A + B = C • addition reactions: a general type of reaction in which a substance adds to the multiple bond of an unsaturated reactant to yield a saturated product that has only single bonds. • 2 reactants add together to form a single product. • Most reactions of carbon-carbon multiple bonds are addition reactions. H H H H C=C + H2 H – C – C – H H H H H ethylene ethane
2. Elimination Reaction A B + C • elimination reactions: a general reaction type in which a saturated reactant yields an unsaturated product by losing groups from two adjacent carbons • Eliminations occur when a single reactant splits into two products H OH H2SO4 H H H – C – C – H C=C + H2O H H H H ethanol ethylene
3. Substitution Reactions AB + C AC + B • substitution reactions: a general reaction type in which an atom or group of atoms in a molecule is replaced by another atom/group of atoms • Occurs when two reactants exchange parts to give 2 new products. H H H – C – H + Cl – Cl H – C – Cl + HCl H H methane chloromethane
4. Rearrangement Reaction A B • rearrangement reactions: a general reaction type in which a molecule undergoes bond reorganization to yield an isomer CH3 CH3H2SO4 CH3 H C=C C=C H H H CH3 cis-2-butene trans-2-butene
Classify each of the following as an addition, elimination, substitution, or rearrangement. • CH3Br + NaOH CH3OH + NaBr substitution • H2C=CH2 + HCl CH3CH2Cl addition • CH3CH2Br CH2=CH2 + HBr elimination
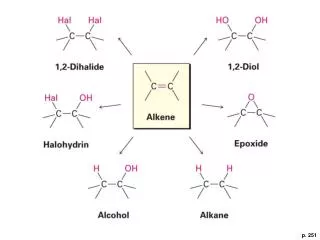
1. Addition Reactions • hydrogenation • halogenation • hydration
Hydrogenation • hydrogenation: the addition of H2 to a multiple bond to give a saturated product alkene (or alkyne) + H2 (g) alkane • used commercially to convert unsaturated vegetable oils, which contain numerous double bonds, to the saturated fats used in margarine and cooking fats. metal catalyst
Example + H2Pd catalyst 1-methylcyclohexene methylcyclohexane H H CH3 H CH3 H
Pd Pd Practice Give the structure of the products: CH3CH2CH=CH2 + H2 =CH2 + H2CH3 CH3CH2CH2CH3
Your Turn • What product would you obtain from: CH3CH2CH2CH=CHCH3 + H2 CH3CH2CH2CH2CH2CH3 Pd
Halogenation • halogenation: the addition of F2, Cl2, Br2, or I2 to a multiple bond to give a dihalide product. H H H H C=C + Cl2 H – C – C – H H H Cl Cl ethylene 1,2 dichloroethane
Practice • What products would you expect from the following halogenation reactions? • 2-methylpropene + Br2 CH3 CH3 CH2=CCH3 + Br2 BrCH2CCH3 Br 1,2-dibromo-2-methylpropane • 1-pentene + Cl2 CH2=CHCH2CH2CH3 + Cl2 ClCH2CHCH2CH2CH3 Cl 1,2-dichloropentane
Your Turn • What product would you expect from: CH3 CH3CH2CH=CCH2CHCH3 + Cl2 ? CH3 Cl CH3 CH3CH2CHCCH2CHCH3 Cl CH3 4,5-dichloro-2,4-dimethylheptane
Hydrohalogenation • hydrohalogenation: the addition of HF, HCl, HBr, or HI to a multiple bond to give an alkyl halide product H H H H C=C + HCl H – C=C – H = CH3CH2Cl H H H Cl HF < < HCl < HBr < HI increasing rate of addition
CH3 H CH3 C=C + HBr CH3 – C – CH3 CH3 H Br 2-methylpropene 2-bromo-2-methylpropane (sole product) But why not CH3 CH3 – C – CH2 ? H Br
Markovnikov’s Rule • In the addition of HX (X = a halogen) to an alkene: • the H attaches to the carbon atom that already has the most H’s • the F, Cl, Br, or I atom attaches to the carbon that has the fewer H
Practice • What product is expected? CH3 CH3CH2C=CHCH3 + HCl ? CH3 CH3CH2CCH2CH3 Cl 3-chloro-3-methylpentane
Your Turn • What products do you expect from… 1-hexene + HBr CH3 CH3CHCH=CH2 + HCl CH3CHCH2CH2CH2CH3 Br CH3 CH3CHCHCH3 Cl
Hydration • hydration: the addition of water to a multiple bond to give an alcohol product • An alkene doesn’t react with water if the two are just mixed together. • You need the right conditions, such as in the presence of a strong acid catalyst, for a hydration reaction to occur. H H H2SO4 H H C=C + H2O H – C – C – H H H H OH ethylene ethyl alcohol
Markovnikov’s Rule Still Applies • as before, Markovnikov’s rule can be used to predict the product when water adds to an alkene • the H adds to the carbon with the more hydrogens • the OH adds to the carbon with the fewer hydrogens CH3H2SO4 CH3 C = CH2 + H2O CH3CCH3 CH3250 °C OH 2-methyl-2-propanol
CH3 OH Practice • What products do you expect from: H2SO4 =CH2 + H2O ? CH3H2SO4 CH3C = CH + H2O ? CH3 CH3 CH3C CH2CH3 OH
Your Turn • Write the reaction of 2-methyl-2-pentene with water in the presence of an acid catalyst. OH CH3C = CHCH2CH3 + H2O CH3CCH2CH2CH3 CH3 CH3 H2SO4
Arenes/Aromatic Compounds • aromatic (arene): the class of compounds containing benzene-like rings • the simplest aromatic compound is benzene (C6H5) • often represented as a six-member ring with 3 double bonds, but this doesn’t fully account for its chemical behavior.
Benzene • Benzene and other aromatic compounds are much less reactive than alkenes. • Benzene’s relative lack of chemical reactivity is a consequence of its structure.
Structure of Benzene • All 6 C-C bonds in benzene are identical. • Benzene’s true structure is an average of the two. The electrons in the double bonds are not held between specific atoms but are free to move over the entire ring. • resonance: the phenomenon where the true structure of a molecule is an average among two or more conventional structures +
Aromatic Compounds • Aromatic compounds are not limited to those with rings only containing carbon. • Many aromatic compounds contain nitrogen. • If there is a six-membered ring in which double-bond electrons are equally shared around the ring, it is classified as aromatic. pyridine indole adenine