Acid-Base Chemistry
Acid-Base Chemistry. CDT Touafek CDT Johnson. Equilibria of Acid-Base Buffer Systems. Acid-Base buffer: Solution that lessens changes in [H30+] resulting from the addition of acid or base.


Acid-Base Chemistry
E N D
Presentation Transcript
Acid-Base Chemistry CDT Touafek CDT Johnson
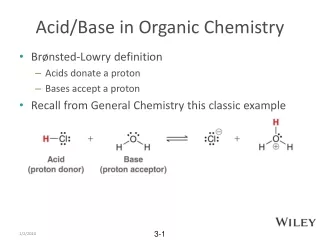
Equilibria of Acid-Base Buffer Systems • Acid-Base buffer: Solution that lessens changes in [H30+] resulting from the addition of acid or base. • Common-ion effect: Shift in position of ionic equilibrium away from ion involved in in chemical process. • Key equation: Henderson-Hasselbalch equation.
Acid-Base Titration Curves • Equivalence point: Point in titration when the number of moles of species added is stoichometrically equivalent to the original moles of the other species. • Titration: Method of determining the concentration of a solution by monitoring its reaction with a solution of known concentration.
Sample Problem for Acid-Base Reactions What is the pH of a buffer consisting of 0.34 M HSO3- and 0.78 M SO3ˆ2- (pKb of SO3ˆ2- = 4.32) HSO3-(aq) + H2O(l) SO3ˆ2-(aq) + OH-(aq) base = [0.34] acid = [0.78] pKa + pKb = 14 pKa= 14 – 4.32 pKa = 9.68 pH=pKa + log([base]/[acid]) pH= 9.68 + log([0.34]/[0.78]) pH = 9.1035 9.10