Implementing Global Data Standards for UDI Compliance in Manufacturing: Cook's Journey
Explore how Cook, with 9 manufacturing companies and 10 clinical divisions in 135 countries, achieved UDI compliance by sorting products, forming teams, and implementing changes like labeling and data verification.

Implementing Global Data Standards for UDI Compliance in Manufacturing: Cook's Journey
E N D
Presentation Transcript
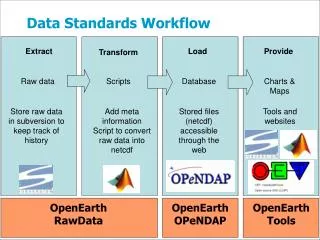
Data Standards Implementation Chuck Franz Vice President & Chief Information Officer IMDMC Annual Conference Indianapolis, November 7, 2013
Cook overview • 9 manufacturing companies • 10 clinical divisions • Coverage in 135 countries • 16,000 SKUs
Final steps to UDI compliance • Sort products by classification • Identify gaps • Form cross-functional teams working on operations and systems • Communicate / Educate
Our plan to become compliant • Labeling • Change date format • Label redesign • Alternative AIDC • GUDID • Data collection, verification, validation • Approval / submission process • Quality systems • Conforming amendments • Device history record • Direct part marking • Evaluate technologies • Validate manufacturing processes • Integrate into manufacturing flow and IT