REVERSE OSMOSIS


REVERSE OSMOSIS
E N D
Presentation Transcript
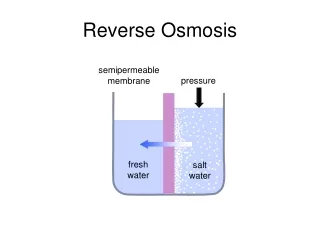
When pure water and a salt solution are introduced on opposite sides of a semipermeable membrane in a vented container, the pure water diffuses through the membrane and dilutes the salt solution, At equilibrium, the liquid level on the saline water side of the membrane will be above that on the freshwater side; this process is known as osmosis. REVERSE OSMOSIS
The effective driving force responsible for the flow is osmotic pressure. This pressure has a magnitude dependent on membrane characteristics, water temperature, and salt solution properties and concentration. By applying pressure to the saline water, the flow process through the membrane can be reversed. When the applied pressure on the salt solution is greater than the osmotic pressure, fresh water diffuses in the opposite direction through the membrane and pure solvent is extracted from the mixed solution; this process is termed reverse osmosis (RO).
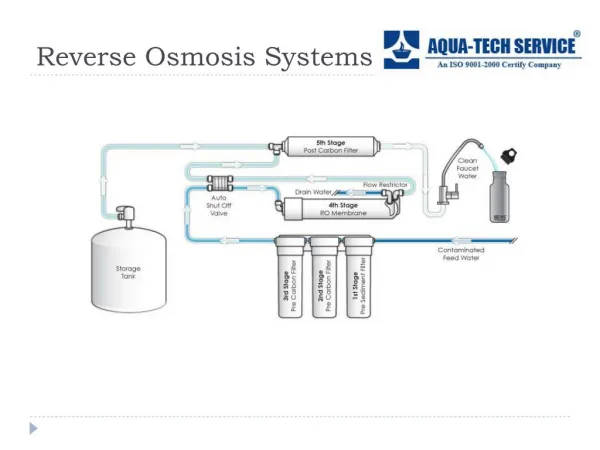
The feed stream is split into two fractions - a purified portion called the product water (or petmeate) and a smaller portion called the concentrate' containing most of the impurities in the feed stream. At the far end of the feed-water passage, the concentration (dewatered) reject stream exits from the cell. After permeating the membrane, the product (fresh-water) flow is collected. The fundamental difference between reverse osmosis and electrodialysis is that in reverse osmosis the solvent permeates the membrane, while in electrodialysis the solute moves through the membrane.
The percentage of product water obtained from the feed stream is termed the recovery, typically around 75 percent. The ratio (F-P)/F or the concentration of a solute species in the feed (F) minus that in the product (P) over the concentration in the feed, is called the rejection of that species. Solids rejection depends on factors such as types and forms of solids, membrane types, recovery, pressure, and pH. Cellulose acetate is a common membrane material, but others include nylon and aromatic polyamides.
for purification purposes we employ the reverse of osmotic flow. For this to happen, we need to apply external pressure in excess of osmotic pressure. The osmotic pressure is given by: Of course, you should be familiar with this equation (the Ideal Gas Law), where 'n' is the molar concentration of solute, R is the universal gas law constant, and T is absolute temperature in °K. The permeate flow can be calculated from: is the membrane permeability coefficient
DISINFECTION Disinfection is a unit process used both in water and wastewater treatment. the process used to control waterborne pathogenic organisms and prevent waterborne disease is called disinfection. The most commonly used disinfectants and oxidants (in no particular order) are chlorine, chlorine dioxide, chloramines, ozone, and potassium permanganate.
Desired characteristics of a disinfectant include the following: 1. It must be able to deactivate or destroy any type or number of disease-causing microorganisms that may be in a water supply, in reasonable time, within expected temperature ranges, and despite changes in the character of the water (e.g., pH). 2. It must be nontoxic. 3. It must not add unpleasant taste or odor to the water. 4. It must be readily available at a reasonable cost and be safe and easy to handle, transport, store, and apply. 5. It must be quick and easy to determine the concentration of the disinfectant in the treated water. 6. It should persist within the disinfected water at a high enough concentration to provide residual protection through the distribution.
The factors affecting the efficiency of disinfection are • Type, condition and concentration of organisms to be destroyed. • Type and concentration of disinfectant • Contact time and concentration of disinfectants in water and • Chemical and physical characteristics of water to be treated particularly the temperature, pH and mineral constituents.
CHLORINATION The addition of chlorine or chlorine compounds to water is called chlorination. Chlorination is considered to be the single most important process for preventing the spread of waterborne disease. In water chlorine hydrolyzes to form hypochlorous acid (HOCL), as shown by the following reactions:
The hypochlorous acid undergoes further ionization to form hypochlorite ions (OCl-) : Equilibrium concentrations of HOCl and OCl depend on the pH of the wastewater. Increasing the pH shifts the preceding equilibrium relationships to the right, causing the formation of higher concentrations of HOC1. Chlorine may also be applied as calcium hypochlorite and sodium hypochlorite. Hypochloritesare salts of hypochlotous acid.