BIOMATERIALS sensu stricto
280 likes | 526 Vues
BIOMATERIALS sensu stricto. Titanium and titanium based alloys bioceramics: Alumina, Zirconia Carbon Hydroxyapatite glasses (vetroceramics, bioglasses). Titanium and Titanium-based alloys. Titanium : a light (AW 47,9), non-magnetic metal.

BIOMATERIALS sensu stricto
E N D
Presentation Transcript
Titanium and titanium based alloys • bioceramics: • Alumina, • Zirconia • Carbon • Hydroxyapatite • glasses (vetroceramics, bioglasses)
Titanium: a light (AW 47,9), non-magnetic metal. The ninth most common element in the earth crust (after oxygen, silicon, aluminum, iron, magnesium, calcium, sodium, potassium), constituting the 0,6% and also the fourth most abundant structural metal after aluminum, iron and magnesium. As the ionic radius of titanium is close to that of other common elements (Al3+, Fe3+, Mg2+), most minerals, stones and soil contain small amounts of titanium, although proper titanium minerals, containing more than 1 % of titanium are only found in few places.
Mineral sources of titanium are rutile (TiO2) and ilmenite (FeTiO3), more common. Rutile contains 90-97% of TiO2, together with impurities of silicon, iron, vanadium, niobium and tantalium.
Two treatments of the minerals. One, the Kroll process, uses chlorine to yield TiCl4, then reduced with metallic Na and Mg to spongy metallic Ti. TiCl4 + 4 Na Ti + 4 NaCl The other in the electrochemical reduction of TiO2 in a CaCl2 bath. By successive melting processes metallic titanium is purified (at high temperature Ti combines with O2, N2, H2, C, Fe, therefore its melting is delicate). Titanium in standard conditions is a silvery metal with low density (4,54 g/cm3), high mechanical strength (660 MPa) and marked resistance to corrosion.
Most used alloys in the biomedical field are: • ASTM F67 (nearly-pure Titanium: 98,9 - 99,6 %) used in dentistry or as coating (poor mechanical properties) • ASTM F136 (Ti-6Al-4V), used in orthopedics (where high loads are applied), because of good mechanical properties, resistance to corrosion, biocompatibility, and elastic modulus closer to that of the bone than other alloys.
ASTM F67 As “commercially pure” Ti actually contains impurities, ASTM (American Society for Testing and Materials) uses a ranking into 4 groups (grade 1, 2, 3 and 4), for each of which the highest tolerable content in N, C, O and Fe, as well as the minimum values of some mechanical properties.
The four grades of commercially pure (CP) Ti differ in oxygen content and mechanical properties Grade 1: CP Ti with a low oxygen content: low tensile strength , high ductility. Grade 2: CP Ti with a higher oxygen content: the most used species with best compromise between the various mechanical requiements. Grado 3: CP Ti used for pressure vessels. Grado 4: Ti “commercially pure” for aeronautics
Pure Ti has a phase transition di fase at 882°C from Ti-α (HCP) to Ti-β(BCC), stable up to the melting point (1668 °C). Alloying with other elements stabilizes either Ti-α or Ti-β. Aluminum stabilizes Ti-α (increased temperature of transition) V has the opposite effect
882ºC Content in Al of F136
Aim of alloying is to increase mechanical properties. Alloys are termed as Ti-α, Ti-(α+β) and Ti-β according to the phase present at room temperature. As the BCC structure of Ti-β has more slide planes than the HCP Ti-α, Ti-β is more easily workable. Alloys β and (α + β) are formed at high temperatures
ASTM F67 • Being nearly pure Ti, it is monophasic of type a (HCP) • grain size 10- 150 mm. • interstitial atoms (C, N, O) causes strengthening. • The surface TiO2 layer brings about resistance to corrosion and to good osteointegration
ASTM F136 • An a-b alloy at ambient temperature • its microstructure depends heavily on thermal history. • If heated between 700 and 950°C (below the transition to b): tiny grains of α(3-10 micron) with crystals of Ti-b at the boundaries of the primary a phase. • This type of structure is recommended for surgical implants
If heated above 975°C and then slowly cooled to ambient temperature, a biphasic structure is obtained, where Ti.a HCP (rich in Al and poor in V) precipitates within the grains of the Ti-b BCC matrix, as sotto forma di lamellae or oriented needles. If cooling is quick, a microstructure is obtained due to solid-state non-diffusive transformations (martensitic).
Metallographic micrographies concerning different microstructures A: lamellar grains of HCP Ti- plus BCC Ti-; B: granular structure; C: globular structure. A B C
Cracks induced by cyclic loads (fatigue tests) originate from grain boundaries between Ti-a and Ti-b. • Microstructures with a grains small (<20 micron) and round (not needle-like or lamellar) and a low interfacial area a/b show higher fatigue limit (500-700 MPa), because they stand better crack formation. • Lamellar structures with high a/b interfacial area have fatigue limit 300-500 MPa. Treatments with hydrogen further improve the mechanical properties: Yield Strength: 974-1119 MPa, UTS: 1025-1152 MPa, Fatigue limit: 643-669 MPa
BIOCOMPATIBILITY • Very high, due to the formation of a surface layer of Titanium oxide, chemically inert (passivation) • Extensive use in implants. • (Dental alloys may cause allergies) • for the same reason: i) good resistance to corrosion; ii) lack of taste (no metallic aroma)
Further favorable features: RADIOTRASPARENCY: A prosthesis in Ti may be X-rayed to control the integrity of a piece THERMAL CONDUCTIVITY: 14 times less than Au. No gun pulp irritation of thermal origin as with Au alloys
Note the better mechanical properties of F136 as compared to F67
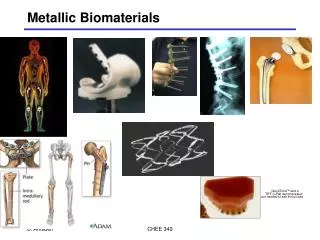
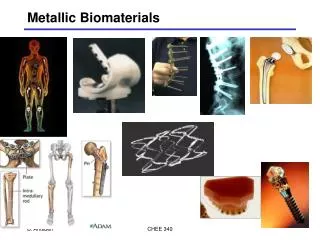
Use of Ti for implants (all main joints: hip, knee, elbow, shoulder)