HEAT ENERGY
HEAT ENERGY. What is HEAT? Form of energy and measured in JOULES Particles move about more and take up more room if heated – this is why things expand if heated It is also why substances change from: solids liquids gases when heated.

HEAT ENERGY
E N D
Presentation Transcript
HEAT ENERGY What is HEAT? Form of energy and measured in JOULES Particles move about more and take up more room if heated – this is why things expand if heated It is also why substances change from: solids liquids gases when heated Visit www.worldofteaching.com for more free powerpoints
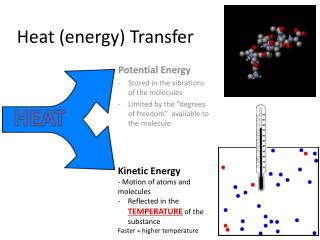
Heat and Temperature • The temperature of an object tells us how HOT it is • Measured in degrees Celsius - °C • It is NOT the same as heat energy although the two quantities are related. e.g. a beaker of water at 60 °C is hotter than a bath of water at 40 °C BUT the bath contains more joules of heat energy
Heating and Cooling • If an object has become hotter, it means that it has gained heat energy. • If an object cools down, it means it has lost energy
Heating and Cooling cont… • Heat energy always moves from:HOT object COOLER object e.g.Cup of water at 20 °C in a room at 30°C - gains heat energy and heats up – its temperature rises Cup of water at 20 °C in a room at 10°C loses heat energy and cools down – its temperature will fall.
Temperature • There are two common temperature scales. • On the Fahrenheitscale, water freezes at 32 degrees and boils at 212 degrees. • The Celsius scale divides the interval between the freezing and boiling points of water into 100 degrees.
Solving Problems A friend in Paris sends you a recipe for a cake. The French recipe says to bake the cake at a temperature of 200 °C for 45 minutes. At what temperature should you set your oven, which uses the Fahrenheit scale?
Converting to Kelvin • The Kelvintemperature scale is useful in science because it starts at absolute zero. • To convert from Celsius to Kelvin, you add 273 to the temperature in Celsius. • K = °C + 273 • °C = K-273
HEAT ENERGY Energy transfer Conduction Convection Radiation
Conduction • Heat is transferred through a material by being passed from one particle to the next • Particles at the warm end move faster and this then causes the next particles to move faster and so on. • In this way heat in an object travels from:the HOT end the cold end
Conduction cont… • Occurs by the particles hitting each other and so energy is transferred. • Can happen in solids, liquids and gases, • Happens best in solids-particles very close together • Conduction does not occur very quickly in liquids or gases
Conductors • Materials that conduct heat quickly are called conductors • All metals are good conductors of heat • Copper is a very good conductor of heat • Pans for cooking are usually made with a copper or aluminium bottom and plastic handles
Insulators/poor conductors • Materials that conduct heat slowly or poorly are called insulators • Glass, wood, plastic and rubber are poor conductors (good insulators) • Nearly all liquids including water are poor conductors (good insulators) • Gases, including air are poor conductors,e.g., wool feels warm because it traps a lot of air • A fridge has insulation material round it to keep it cold – reduces amount of heat conducted to inside from the warmer room
Conductor or Insulator? • Wood? • Aluminium? • Plastic? • Glass? • Iron? • Polystyrene? • Copper? • Cardboard?
Convection • Takes place in material where particles can move around inside the material, i.e. liquid or gas • The heat is carried by the particles themselves moving Convection currents • Occur because an area with warm particles expands and becomes less dense than the cooler areas nearby. The warm area rises.Cooler particles fall into the space left by the warm particles and convection current is set up
Convection Currents • Hot liquids and gases expand and rise while the cooler liquid or gas falls 2. Goes across 3. Then down 1.Hotair rises 4. And across
Convection cont… • The sun can cause large convection currents - WINDS • During daytime the land warms up more than the sea. The warm air rises over the land and cool air falls over the sea. So we feel a sea breeze. • Rising convection currents can be uses by glider pilots to keep their planes in the air and by birds to stay aloft.
Radiation • Transfer of heat directly form the source to the object by a wave, travelling as rays. • Heat radiation is also known as • All objects that are hotter than their surroundings give out heat as infra-red radiation • Heat transfer by radiation does not need particles to occur and is the only way energy can be transferred across empty space INFRA-RED RADIATION
Emitters • Hotter objects emit (give out) heat • Different surfaces emit heat at different speeds • A dull black surfaces loses energy more quickly – it is a good radiator • A bright shiny or white surface is a poor radiator • Marathon runners need to keep warm at the end of races, covering in shiny blankets reduces radiation and therefore heat loss.
Emitters of heat Bright shiny canPoor radiator Dull black canGood Radiator
Absorbers • Cooler objects absorb (take in) heat • Substances absorb heat at different speeds • Dull, black surfaces absorb heat quickly • Bright, shiny surfaces absorb heat slowly • In hot countries, people wear bright white clothes and paint their houses white to reduce absorption of energy from the sun. • Petrol storage tanks sprayed silver to reflect sun’s rays
Absorbers Shiny, bright canPoor absorber Dull black canGood absorber
Key Words Temperature Cold Insulator Radiation Heat Transfer Convection Conduction Conductor Absorber Emitter
Heat Transfer QUIZ • Microwaving method • Warmth from a fire • Accidentally touching a hot light bulb • Sunbathing on the beach • Noodles boiling in a pot • “Nuking” your hot chocolate • Burning your finger as you remove it from the microwave • A pot sitting on a HOT stovetop eye