Diffusion & Osmosis
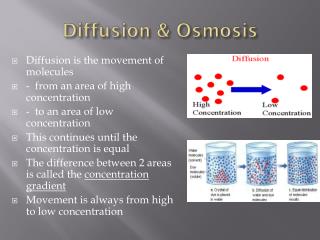
Diffusion & Osmosis. Diffusion is the movement of molecules - from an area of high concentration - to an area of low concentration This continues until the concentration is equal The difference between 2 areas is called the concentration gradient

Diffusion & Osmosis
E N D
Presentation Transcript
Diffusion & Osmosis • Diffusion is the movement of molecules • - from an area of high concentration • - to an area of low concentration • This continues until the concentration is equal • The difference between 2 areas is called the concentration gradient • Movement is always from high to low concentration
Diffusion (Single celled organisms) • Single celled organisms use up oxygen & food • & produce CO2 & waste • There is generally a gradient between the inside & outside of the cell • The cell membrane allows oxygen, CO2 food across freely (freely permeable) • Oxygen/food will diffuse into the cell • CO2/waste will diffuse out of the cell
Food diffusion -single celled organisms • Single celled animals (e.g. Amoeba) engulfs food by phagocytosis • Food becomes engulfed in a food vacuole • Food is digested by enzymes (inside lysosomes) • Inside the vacuole there is a high conc. of food products, a low conc. in the cytoplasm • The food diffuses from high conc. to low conc.
Multicellular organisms • In animals • Diffusion is important for exchange of gases • Blood returning to lungs • - high conc. of CO2, low conc. of oxygen • CO2 diffuses out of blood into air sacs • Oxygen diffuses from air sacs into blood • Diffusion also important for dissolved food & wastes • In plants • - CO2 diffuses from the air into the leaves • - water diffuses from the soil into the roots
Role of Cell Membrane • Cell membrane is semi-permeable • Allows small molecules (e.g. water, oxygen) to pass across freely • Won’t allow large molecules (e.g. starch) to pass across • Membranes have tiny pores or gaps
Water Concentrations • A solution with the higher water conc (HWC) is said to be HYPOTONIC • The solution with the lower water conc (LWC) is HYPERTONIC • If the 2 solutions are of equal conc, they are ISOTONIC • Molarity (M) is the conc of solute (e.g. sugar/salt) dissolved in water
Effect of osmosis on cells • If a solution outside a cell is hypotonic, water will diffuse in • The cell will expand • Animal cells will eventually burst • Plant cells will swell and become hard - TURGID • If a solution outside the cell is hypertonic, water will diffuse out • The cell will shrink & shrivel up • Plant cells are said to be PLASMOLYSED
Osmosis & Cells • In plants, useful for: • - Soil to roots (water into plant) • - Xylem to stem (for stem support) • - xylem to green leaves (for photsynthesis) • In single celled organisms: • e.g Paramecium • contractile vacuoles control water levels (OSMOREGULATION)