Understanding Moles in Chemistry: Basics and Calculations
This guide introduces the concept of moles in chemistry, essential for measuring substances in reactions. A mole is defined as 6.022 x 10^23 particles, equivalent to the number of atoms in 12 grams of Carbon-12. We will explore how to calculate moles using the formulas Moles of Atoms = Mass / Relative Atomic Mass (R.A.M.) and Moles = Mass / Relative Formula Mass (R.F.M.) for compounds. Several examples, including calculations for elements like carbon, copper(II) oxide, and various molecules, will help solidify your understanding of moles.

Understanding Moles in Chemistry: Basics and Calculations
E N D
Presentation Transcript
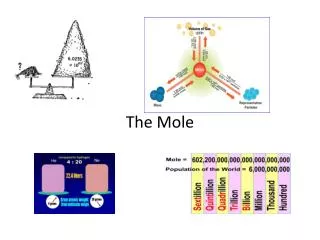
Toosmalltosee • Becauseatoms are so small, itmakes more sensetomeasurelargeamounts of atoms in reactions. • One mole of a substancecontainsthesamenumber of particles as atoms in 12 grams of Carbon-12
TheAvagadroConstant • 1 mole of a substancecontains 6.022 x 1023particles • Thats 602 200 000 000 000 000 000 000 particles!!!
Equation • Tocalculatethe moles in a substancewe use thefollowingequation: • Moles of Atoms = Mass/RelativeAtomicMass
R.A.M. • RelativeAtomicMassisthe Ar valuefromthePeriodicTable
Question • Howmany moles of atoms are there in 2.4 g of carbon?
Answer • Moles of atoms = mass/R.A.M. • = 2.4/12 = 0.2 moles
TheMagicTriangle • Usingthemagictrianglewe can rearrangetheequationtofindmassinstead of moles
Question • Whatisthemass of 0.1 moles of CarbonAtoms?
For a compound • A compound has more thanonetype of atom, so wehavetofindtheRelative Formula Mass (R.F.M.) • We do thisbyaddingthe Ar of theelements in themolecule. • So theequationthenbecomes: • Moles = Mass/R.F.M.
Question • Howmany moles are there in 8 g of copper(II) oxide (formula CuO)? • 1. Findthe RFM Cu = 64 O = 16 So the RFM of CuOis 80 ….thenjustfollowtheequation…
Questions • Howmany moles of atoms are there in: • 2 g of hydrogen • 36 g of carbon • 160 g of oxygen • 14 g of nitrogen • 0.19 g of flourine
Questions • Howmany moles of molecules are there in: • 26 g of H2O • 170 g of NH3 • 1.6 g of CH4 • O.2 g of C2H6 • 16 g of NH4NO3