Ch 9 (Part 3): E.T.C./ Oxidative Phosphorylation
300 likes | 547 Vues
Ch 9 (Part 3): E.T.C./ Oxidative Phosphorylation. So far, in glycolysis & the Krebs cycle, 1 glucose molecule has resulted in: 4 ATPs (2 from glycolysis, 2 from Krebs) 10 NADH (2 from gly., 2 from acetyl-CoA step, 6 from Krebs Cycle) 2 FADH 2 ( from Krebs Cycle ). x2.

Ch 9 (Part 3): E.T.C./ Oxidative Phosphorylation
E N D
Presentation Transcript
So far, in glycolysis & the Krebs cycle, 1 glucose molecule has resulted in: • 4 ATPs (2 from glycolysis, 2 from Krebs) • 10 NADH (2 from gly., 2 from acetyl-CoA step, 6 from Krebs Cycle) • 2 FADH2 (from Krebs Cycle) x2
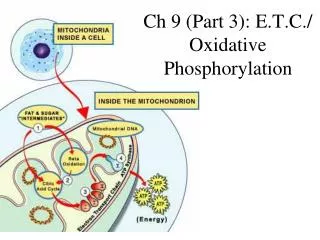
ELECTRON TRANSPORT CHAIN (E.T.C.) • a collection of molecules embedded in the inner membrane of mitochondrion (folding of inner membrane to form cristae)
the groups along the chain alternate between reduced & oxidized states as they accept and donate electrons • each successive group is more electronegative than the group before it, so the electrons are “pulled downhill” towards OXYGEN (the final electron carrier!)
as molecular oxygen (O2) is reduced, it also picks up H+ from the environment to form water(H2O)
ATP Production of the E.T.C. Typically, the ATP produced is as follows: 1 NADH 3 ATP 1 FADH2 2 ATP (FADH2 is “dropped off” at a lower point in the E.T.C., so it generates fewer ATPs) (“exchange rate”)
The E.T.C. does not make ATP directly – it generates a proton gradient across the mitochondrial membrane, which stores potential energy that can be used to phosphorylate ADP • Some of the proteins in the e.t.c. pump H+ against their gradient…this generates a proton (H+) gradient called the PROTON MOTIVE FORCE.
H+ (outer matrix) H+ H+ -protons then diffuse back across the membrane through the ATP synthase complex which causes the phosphorylation of ADP to form ATP! H+ H+ ADP + Pi ATP (inner matrix)
CHEMIOSMOSIS: • the coupling of exergonic electron flow down an E.T.C. to endergonic ATP production; the proton gradient drives ATP synthesis as protons diffuse back across the membrane **accomplished by ATP synthase complex! We Have an Animation!!
SUMMARY: • most ENERGY flows in this sequence: Glucose NADH e.t.c. proton ATP motive force
PROCESS ATP produced by subs. Phos. Reduced coenz. ATP produced by oxid. phos. (in the E.T.C.) TOTAL Glycolysis 2 ATP 2 NADH 4-6 ATP 6-8 ox. of pyruvate to acetyl CoA (LINK) 2 NADH 6 ATP 6 Krebs cycle 2 ATP 6 NADH 2 FADH2 18 ATP 4 ATP 24 TOTAL 36-38!
Energy in organic compd. NADH/FADH2 ETC (oxidative phosphorylation)
• approximately 40% of energy in glucose is converted to ATP • the remaining energy is lost as heat
Alternative Metabolic Pathways - Vocabulary: • aerobic: existing in presence of oxygen • anaerobic: existing in absence of oxygen • fermentation = anaerobic catabolism of organic nutrients
Alcoholic Fermentation Pyruvate + NADH ethanol + CO2 + NAD+ • pyruvate is converted to ethanol • NADH is oxidized to NAD+ (recycled) • performed by yeast and some bacteria
Lactic Acid Fermentation Pyruvate + NADH lactic acid + NAD+ • pyruvate is reduced to lactic acid • NADH is oxidized to NAD+(recycling of NAD+) • performed by some bacteria (producing cheese and yogurt) • performed by our muscle cells when oxygen is scarce
Fermentation differs from cellular respiration in: • method NADH is recycled back to NAD+ • in fermentation, the final electron acceptor is pyruvate, not O2 • amount of total energy harvested (2 ATP from glycolysis instead of 36-38 using Kreb’s and ETC)
EXTRA VOCABULARY: E. Coli • Obligate anaerobes: only grow in absence of oxygen (e.g. Clostridium botulinum) • Obligate aerobes: only grow in presence of oxygen Micrococcus luteus
Facultative anaerobes: can grow in either presence or absence of oxygen (e.g. yeast; bacteria that make yogurt, cheese; our muscle cells at the cellular level)
*pyruvate is a “fork” in the metabolic road which leads to 2 alternate catabolic routes: -if O2 is present: Krebs and e.t.c. -if no O2 is present: Fermentation