Nomenclature
Nomenclature. Unit 6 - Bonding. Types of Ions. Monatomic = 1 atom Representative elements TYPICALLY have only one possible charge Names Cations = name of the element Ca 2+ = Calcium ion Anions = remove ending, add “–ide” N 3- = Nitride. Types of Ions.


Nomenclature
E N D
Presentation Transcript
Nomenclature Unit 6 - Bonding
Types of Ions • Monatomic = 1 atom • Representative elements TYPICALLY have only one possible charge Names • Cations = name of the element • Ca2+ = Calcium ion • Anions = remove ending, add “–ide” • N3- = Nitride
Types of Ions • Transition metals and some representative elements have more than one charge. • There are two ways to name these elements…
Types of Ions • Stock System • Element name with charge in parentheses (Roman Numerals) • Copper (I) = Cu+ • Latin System • Latin root + suffix • - ous = Lower charge • - ic = Higher charge • Ferric and Ferrous
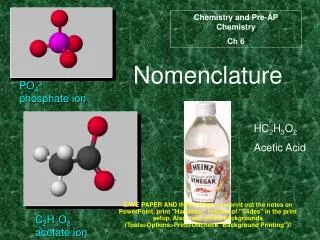
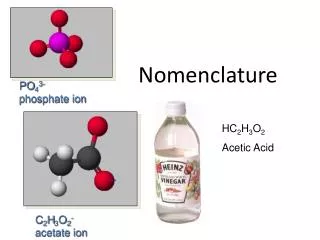
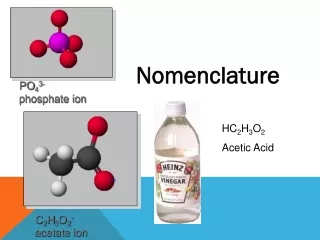
Types of Ions • Polyatomic Ions = A charged GROUP of bonded atoms • Acts as ONE UNIT in an ionic bond • Cations: Only NH4+ • Anions: More common as polyatomics • Look at the back of the periodic table!!
Naming Ionic Compounds • Cation is always named first (No # prefixes) • Cation take the name of the element • Transition metals use the anion to find the charge of the cation • (Roman numeral = charge) • Anion • Monatomic end in –ide • Polyatomic name from chart
Naming • Practice…..Practice…..Practice • CaF2 • calcium fluoride • Li2S • Lithium sulfide • FeCl2 • Iron (II) chloride • FeCl3 • Iron (III) chloride
Naming • PtO2 • Platinum (IV) oxide • CuBr • Copper (I) bromide • Zn3N2 • Zinc (II) nitride (Zn is +2) • Sn3P2 • Tin (II) phosphide
Naming • Ternary: • Ca(OH)2 • Calcium hydroxide • NaSCN • Sodium thiocyanate • Al2(CrO4)3 • Aluminum chromate • Mg(NO2)2 • Magnesium nitrite
Naming • NH4Cl • Ammonium chloride • Au2(CO3)3 • Gold (III) carbonate • Pb(CN)4 • Lead (IV) cyanide • FePO4 • Iron (III) phosphate