Bioenergetics
Bioenergetics. Week 2&3. Terms. Metabolism Collection of all chemicals pathways that result in either synthesis (anabolic) or breakdown (catabolic) of molecules Bioenergetics Conversion of “foodstuffs” (fats, carbs, proteins) into a biologically usable form of energy Importance:

Bioenergetics
E N D
Presentation Transcript
Bioenergetics Week 2&3
Terms • Metabolism • Collection of all chemicals pathways that result in either synthesis (anabolic) or breakdown (catabolic) of molecules • Bioenergetics • Conversion of “foodstuffs” (fats, carbs, proteins) into a biologically usable form of energy • Importance: • How can we educate on movement if we don’t know what system is utilized and how it works?! • EPs need to know what system are being utilizing in order to effect change • Help EPs determine why performance is lacking or not possible • Allow EPs to tailor nutritional program with licensed nutritionist
Cellular Chemical Reactions • Muscle fibers convert chemical energy into mechanical energy • Types • Endergenic (endothermic) • Exergenic (exothermic) • Coupled • Most common type in body is oxidation-reduction
Energy and Exercise Chemical Energy (potential energy) Mechanical Energy (kinetic energy) < 25% > 75% Work Heat
ANABOLIC Endergonic Reactions Glucose Glycogen Glycerol + FFAs Triglycerides Protein Amino Acids ADP + CP ATP Exergonic Reactions CATABOLIC
Oxidation and Reduction • Oxidation • Removing an electron • Does not mean oxygen participates, only that it is typically the atom accepting the electron • Reduction • Addition of an electron • Reducing versus Oxidizing agent • Must be coupled because an electron can not be released unless there is an atom who gains it
NAD and FAD • Two important molecules that aid in transfer of electrons • NAD • Nicotinamide adenine dinucleotide • Derived from Niacin (Vitamin B3) • Can only accept one hydrogen • NAD+ and NADH • FAD • Flavin adenine dinucleotide • Derived from riboflavin (Vitamin B2) • Can accept up to 2 hydrogens • FAD+ and FADH
Enzymes • Do not cause a reaction to occur or stop • Do not affect final result of pathway • Regulate the speed/rate of cellular chemical reactions • Classifications • Kinases (add phosphate group) • Dehydrogenases (remove H+) • Oxidases (catalyze O-R reactions with O2) • Isomerases (rearrange atoms to form isomers)
Lock and Key of Enzymes • Unique 3D characteristics which allow regulation of rate • Active sites • Ridges and grooves • Connect to a reactant molecule (substrate) • Create enzyme-substrate complex • Dissociate after reaction occurs but do not dissolve • Ability to work is not constant
Activation energy • Enzymes work by helping to reduce the activation energy • Increase the RATE • Analogies • Reducing obstacle to jump over • Decreasing the size of the hill to climb
Activation energy Activation energy E released E released
Exercise and Enzymes • Why are enzymes important especially when we are exercising?! • Why do we care?! We aren’t biochemists! • What should we be telling our clients about enzymes and their diet?
Factors that Alter Activity of Enzymes • Temperature • Optimum level (36-40 celsius or 96.8-104 farenheit) • Small rise in temperature as with exercise = ? • pH • Optimum level (high 7 to 8) • Increase/decrease in pH alters activity • Exercise = ? • Ceiling with temperature/pH where benefit is lost and activity decreases dramatically
pH and Temperature Effect on Example: “Enzyme A” So if someone is exercising, whether submaximally/maximally/steady state, what does our body use to maintain these systems?
“Foodstuffs” • Fuels for Exercise • Carbohydrates • 4 kcal of energy • Simple (glucose) or Poly (cellulose/starch and glycogen) • Plant or Animal • Fat • 9 kcal of energy • FFA, trigylcerides • Adipose and muscle • Proteins • 4 kcal of energy • How does diet affect a client or athlete’s performance? Why do we care?!
Protein as Energy Source • As long as energy balance and protein intake are adequate, protein will NOT be used as an energy source at rest or during exercise • Why?
Energy • ATP • Adenosine triphosphate • The immediate source of energy for muscular contraction • A high energy phosphate compound • Requires a large amount of E to form • Bonds of ADP + Pi store E
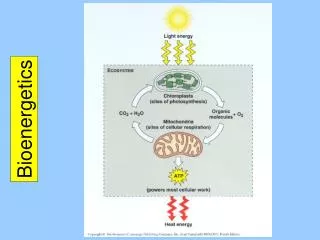
Bioenergetics How do we create energy in the form of ATP during rest and exercise? • Anaerobic • Phosphocreatine system • Glycolysis • Aerobic • Krebs Cycle • Electron Transport Chain
Phosphocreatine (PC) System • PC + ADP ATP + C • Enzymes: ATPase and creatine kinase • Onset of exercise during short-term, high intensity exercise • Less than 5-10 seconds, maximal effort • Examples
PC System • Only a small amount of ATP is “stored” in the cell • The ATP-PC energy system allows for the temporary maintenance of an energy supply (ATP) at the expense of (CP or PC) • Replenishment of PC can only occur during rest from exercise • Since only a limited supply of CP is available in the cell, ATP must be supplied via other energy producing pathways
Work/Exercise ATPase ATP + H2O ADP + Pi + ENERGY ADP + Pi CK ATP + C CP Pi Rest CK ATP + C ADP + CP
Exercise at 150 W Recovery
Creatine Supplement • Individuals with lower initial values of Creatine concentration in muscle have greater increases • Responders and Nonresponders • Evidence: • Short-term loading scheme appears to improve ability to maintain muscular force and power output during exhaustive exercise. • Does not reduce muscle damage from severe exercise • Increases body mass but mostly due to water retention • Negative consequence for athletes needing to “carry their own weight” • No improvement in PC recovery rate post-exercise • Should it be banned? Utilized?
Glycolysis • Anaerobic used to transfer E from glucose/glycogen to make ATP • Net gain of 2 molecules of ATP with glucose / 3 ATP with glycogen • 2 molecules of pyruvate or lactate • Energy investment stage • Energy generation phase • H+ is transported by FAD and NAD+ to be used for later generation of ATP in aerobic pathways • Also considered the first step in the aerobic production of carbohydrates
Critical components • NAD+ and FAD • Essential in order to shuttle away H+ • Reformation of NAD+ occurs • With sufficient O2, shuttled to mitochondria to contribute with aerobic pathways • Without O2, pyruvate takes H+ to form lactate
Video • Anaerobic Energy Systems • Glycolysis
Aerobic ATP Production • Oxidative phosphorylation • 3 steps • Generation of Acetyl CoA • Oxidation of Acetyl CoA (Krebs Cycle) • Electron transport chain
Krebs Cycle – “Citric Acid Cycle” • Acetyl CoA must be created in preparation • 2 Pyruvate from glycolysis • Each molecule of glucose results in 2 turns of Krebs Cycle (2 pyruvate) • Each turn: • 3 molecules of NADH • 1 molecule of FADH • END GOAL: supplies electrons for ETC Each carries electrons in their reduced state
Video • Aerobic Energy Systems • Krebs Cycle • Oxidative Phosphorylation
Electron Transport Chain (ETC) • Occurs in mitochondria • “respiratory or cytochrome chain” • Uses potential E in NADH and FADH to add Pi to ADP forming ATP • Free radicals are formed
Video • Electron Transport Chain
System Fuel O2? Speed ATP Yield Anaerobic ATP-PC system PC No Fastest Very Limited Glycolytic system CHO No Fast Limited Aerobic Oxygen system CHO/ Yes Slow Unlimited Fat / Pro
Case #1 • A competitive weight lifter is attempting to improve his 1-RM for bench press. He is asking you, a EP working at his facility, for advice: • Which system is involved? • What evidence would you provide as an explanation for or against the use of Creatine supplement? • What precautions should you make him aware of?
Case #2 • A female runner is planning on a 10 mile run in preparation for her half marathon in a few months. She is able to run at a steady pace on level or slight inclined ground; however large inclines along the course route give her trouble? What energy system is MOST involved and what can we suggest to her to improve her performance?