Protocol and Case Record Form
DESCRIPTION
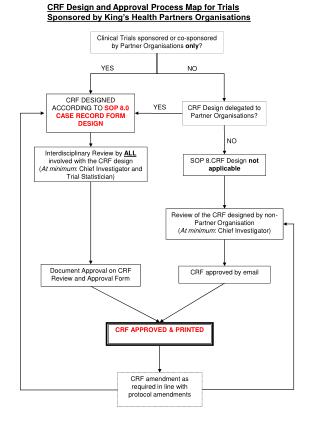
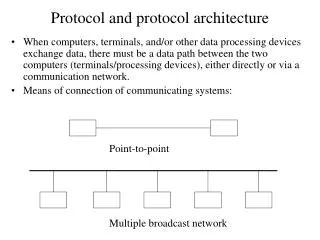
This document outlines the standard protocol and case record form used in clinical trials. It serves as a comprehensive guide for researchers to document participant information, intervention details, and outcome measures accurately. The protocol ensures adherence to regulatory standards and ethical guidelines while collecting data for analysis. Researchers must follow this standardized format to ensure consistency, reliability, and validity of the study findings, facilitating effective communication among stakeholders and contributing to the overall success of the clinical trial.
1 / 30
Télécharger la présentation 

Protocol and Case Record Form
An Image/Link below is provided (as is) to download presentation
Download Policy: Content on the Website is provided to you AS IS for your information and personal use and may not be sold / licensed / shared on other websites without getting consent from its author.
Content is provided to you AS IS for your information and personal use only.
Download presentation by click this link.
While downloading, if for some reason you are not able to download a presentation, the publisher may have deleted the file from their server.
During download, if you can't get a presentation, the file might be deleted by the publisher.
E N D
Presentation Transcript
More Related





















![❤[PDF]⚡ Eyelash Extensions intake & Consultation Form: Lash Client Record book,](https://cdn7.slideserve.com/12911543/eyelash-extensions-intake-consultation-form-lash-dt.jpg)