Isotopes and Mass Number
70 likes | 754 Vues
Isotopes and Mass Number. How Dalton’s atomic theory was wrong: Atoms ARE divisible. (Who is credited with first splitting an atom?) All atoms of a particular element are NOT identical. All atoms of an element do have the same number of protons and electrons, so what could differ?.

Isotopes and Mass Number
E N D
Presentation Transcript
Isotopes and Mass Number • How Dalton’s atomic theory was wrong: • Atoms ARE divisible. (Who is credited with first splitting an atom?) • All atoms of a particular element are NOT identical. • All atoms of an element do have the same number of protons and electrons, so what could differ?
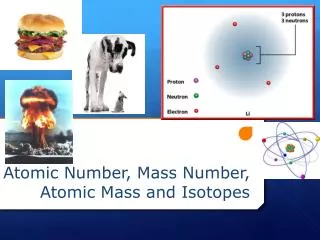
Isotopes and Mass Number (cont.) • Isotopes: atoms with the same number of protons (same element), but with different numbers of neutrons. • Examples: • Potassium (K): K-39, K-40, K-41 • Chlorine (Cl): Cl-35 and Cl-37 • Uranium: U-235 and U-238
Isotopes and Mass Number (cont.) • If atoms have the same number of protons and electrons, but different numbers of neutrons, what else will be different? • Answer: MASS • Mass number = number of protons + number of neutrons
Isotopes and Mass Number (cont.) • Examples: • K-39 = 19 protons + 20 neutrons • K-40 = ___ protons + ___ neutrons • K-41 = ___ protons + ___ neutrons • Isotope symbolic notation: mass number Element Symbol=39K atomic number 19 • Isotope name is potassium-39
Isotopes and Mass Number (cont.) • Review: • Number of protons is the same for all atoms of a particular element • Number of neutrons can vary ( = isotopes) • What is the mass number? • What is the atomic number? • Number of neutrons = ?
Isotopes and Mass Number (cont.) • Number of neutrons = mass number – atomic number • Example: If an atom of neon has an atomic number of 10, and a mass number of 22: • How many protons? Neutrons? Electrons? • What is the NAME of the isotope? • What is the SYMBOL of the isotope?
Practice & Assignment • Practice: p.101 (14) • Study Guide Workbook: pp. 21-22 (13-23)