Enzymes for manipulating DNA
Enzymes for manipulating DNA. *** Buffers and solution conditions*** I. DNA polymerases III. Kinase and alkaline phosphatase IV. Nucleases V. Topoisomerase Course Readings: 19 and 20. Buffers are crucial for activity of enzymes! Ideal biochemical buffers: pKa between 6 and 8

Enzymes for manipulating DNA
E N D
Presentation Transcript
Enzymes for manipulating DNA *** Buffers and solution conditions*** I. DNA polymerases III. Kinase and alkaline phosphatase IV. Nucleases V. Topoisomerase Course Readings: 19 and 20
Buffers are crucial for activity of enzymes! • Ideal biochemical buffers: • pKa between 6 and 8 • Chemically inert • Polar (soluble and not membrane permeable) • Non-toxic • Inexpensive • Salt and temperature indifferent • Tris: pKa is 8.0 • Tris(hydroxymethyl)aminomethane (THAM): the free base for (pH 7.5-8.5) • Tris-HCl: the acidic form (for pH 7-8)
Tris is widely used, but it isn’t perfect: • Buffering is weak below pH 7.5 and above pH 9.0 • pH must be measured using a special pH meter electrode • Toxic to many types of mammalian cell cultures • Tris solution pH changes with temperature! Drops 0.03 pH units for each degree C increase • Tris solution pH changes with concentration! Example: 10mM Tris pH 7.9, 100mM Tris pH 8.0 • Below pH 7.5, use a “Good” buffer: HEPES, Tricine, BES, MOPS, MES
Enzyme “reaction buffers”: • Buffer: Tris, HEPES, etc. • Salt: NaCl, KCl, PO4-, etc.--stabilizes protein structure, facilitates protein-DNA interactions • Divalent metal ions: Mg2+, Ca2+, Zn2+, etc.--often required for enzyme activity • Glycerol: (for storage)--stabilizes protein structure • EDTA: chelates (removes) divalent cations--important especially for storage, if your enzyme is especially sensitive to metal ion-dependent proteases • Beta mercaptoethanol or dithiothreitol: reducing agents that prevent illegitimate disulfide bond formation • Non-specific protein: Bovine serum albumin (BSA) • Other cofactors, eg. ATP, NADH
DNA polymerases--making copies, adding labels, or fixing DNA E. coli DNA polymerase I --the classic DNA polymerase • Moderately processive polymerase • 3'->5' proof-reading exonuclease • 5'->3' strand-displacing (nick-translating) exonuclease • Used mostly for labelling DNA molecules by nick translation. For other purposes, the Klenow fragment is usually preferred
DNA polymerases • Klenow fragment --the C-terminal 70% of E. coli DNA polymerase I; originally prepared as a proteolytic fragment (discovered by Klenow); now cloned • Lacks the 5'->3' exonuclease activity • Uses include: • Labeling DNA termini by filling in the cohesive ends generated by certain restriction enzymes • generation of blunt ends • DNA sequencing
A way of making blunt ended DNA (repair after mechanical fragmentation)
DNA polymerases • Native T7 DNA polymerase --highly processive, with highly active 3'->5' exonuclease • Useful for extensive DNA synthesis on long, single-stranded (e.g. M13) templates • Useful for labeling DNA termini and for converting protruding ends to blunt ends • Modified T7 polymerase (Sequenase) --lack of both 3'->5' exonuclease and 5'->3' exonuclease • Ideal for sequencing, due to high processivity • Efficiently incorporates dNTPs at low concentrations, making it ideal for labeling DNA
DNA polymerases • Reverse transcriptase • RNA-dependent DNA polymerase • Essential for making cDNA copies of RNA transcripts • Cloning intron-less genes • Quantitation of RNA
Reverse transcriptase: The Km for dNTPs is very high (relatively non-processive) Makes a DNA copy of RNA or DNA -- but -- The self-primed second strand synthesis is inefficient “Second-strand” cDNA synthesis is usually done with DNA polymerase and a primer
cDNA library construction using reverse transcriptase cDNA Library Construction Kit (Clontech)
Priming reverse transcriptase: • General RNA amplification: • Oligo(dT)12-18 • Random sequence oligonucleotides • Specific mRNA • Single oligonucleotide sequence complementary to your mRNA NOTE: Reverse transcriptase is error-prone (about 1/500 bp is mutated)
Terminal transferase • template-independent DNA polymerase • Incorporates dNTPs onto the 3' ends of DNA chains • Useful for adding homopolymeric tails or single nucleotides (can be labelled) to the 3' ends of DNA strands (make DNA fragments more easily clonable)
T4 polynucleotide kinase • Transfers gamma phosphate of ATP to the 5’ end of polynucleotides • Useful for preparing DNA fragments for ligation (if they lack 5’ phosphates) • Useful for radiolabelling DNA fragments using gamma 32P ATP as a phosphate donor
alkaline phosphatase • Catalyzes removal of 5’ (and 3’) phosphates from polynucleotides • Useful for treating restricted vector DNA sequences prior to ligation reactions, prevents religation of vector in the absence of insert DNA • Lack of vector 5’ phosphates may inhibit transformation efficiency? Use only when absolutely necessary…
Nucleases • Exonucleases • Remove nucleotides one at a time from a DNA molecule • Endonucleases • Break phosphodiester bonds within a DNA molecule • Include restriction enzymes
Exonucleases • Bal 31 • Double-stranded exonuclease, operates in a time-dependent manner • Degrades both 5’ and 3’ ends of DNA • Useful for generating deletion sets, get bigger deletions with longer incubations
Exonucleases • Exonuclease III--double-stranded DNA • 3’-5’ exonuclease activity • 3’ overhangs resistant to activity, can use this property to generate “nested” deletions from one end of a piece of DNA (use S1 nuclease to degrade other strand of DNA)
Exonucleases • Exonuclease I • 3’-5’ exonuclease • Works only on single-stranded DNA • Useful for removing unextended primers from PCR reactions or other primer extension reactions
Endonucleases • Dnase I • Cleaves double-stranded DNA randomly (also cleaves single-stranded DNA) • Mn++: both strands of DNA cut • Mg++: single strands nicked • Very useful for defining binding sites for DNA binding proteins
DNAse I footprinting Calibrate the nicking: 1 hit per DNA molecule
Drosophila heat-shock factor DNAse I footprinting:Gel following footprinting reaction 0 Sites for interaction of HSF with DNA
Topoisomerase Function: A restriction enzyme and ligase--all in one altering the “linking number” in coiled, constrained (supercoiled) DNA--relaxing DNA twisting during replication Model for function: http://mcb.berkeley.edu/labs/berger/structures.html#modeling
Topoisomerase • Topoisomerase catalyzed ligation is EXTREMELY efficient (>85% of resulting plasmids are recombinant)--excellent for library constructions • Can be used to clone blunt ended DNA (PCR products, restriction digests), T-overhang PCR products (from Taq polymerase), and directional clones • You have to use their plasmid vectors (ie. forget about using your favorite lab plasmid unless you know how to covalently attach topoisomerase)
Enzymes for manipulating DNA *** Buffers and solution conditions*** I. DNA polymerases III. Kinase and alkaline phosphatase IV. Nucleases V. Topoisomerase Course Readings: 19 and 20
Cutting and pasting DNA • Restriction and modification systems • Recognition and cleavage of DNA by restriction endonucleases (REases) • Joining (ligating) DNA molecules • Cloning techniques
Discovery of restriction/modification EOP = efficiency of plating (a measure of phage virulence) = bacteriophage E. coli K has R/M system E. coli C has no M system
Cautions for cloning in E.coli • Strains with methylases (dam or dcm) produce methylated DNA--difficult to cleave with certain enzymes, hard to transform some strains • Strains with restriction systems intact will restrict DNA coming from a host lacking methylases, or from a host with specific types of methylations • Best bet is to delete the restriction systems, but not all cloning strains have this deletion
Types of endonucleases • Type I: multisubunit proteins that function as a single protein complex, usually contain two R subunits,two M subunits and one S subunit • Type II: recognize specific DNA sequences and cleave at constant positions at or close to that sequence to produce 5’-phosphates and 3’-hydroxyls.Most useful in cloning!! • Type III: composed of two genes (mod and res) encoding protein subunits that function either in DNA recognition and modification (Mod) or restriction (Res) • Type IV: one or two genes encoding proteins that cleave only modified DNA, including methylated, hydroxymethylated and glucosyl-hydroxymethylated bases
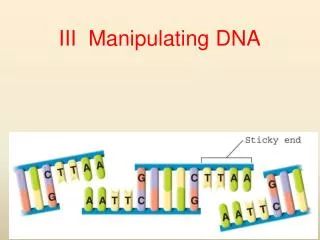
Mode of action of type II REases EcoRI 5´ ... G^A A T T C ... 3´ 3´ ... C T T A A^G ... 5´ EcoRI 5´ ... G^ 3’ 5’ A A T T C ... 3´ 3´ ... C T T A A 5’ 3’ ^G ... 5´
Example recognition sequences for REases 4-cutters: AluI 5´ ... AG^CT ... 3´ blunt ends MspI 5´ ... C^CGG ... 3´ 5’ overhang (2 bp) 6-cutters PvuII 5´ ... CAG^CTG ... 3´ blunt ends KpnI 5´ ... GGTAC^C ... 3´ 3’ overhang (4 bp) 8-cutters NotI 5´ ... GC^GGCCGC ... 3´ 5’ overhang (4 bp) Unusual sites MwoI 5´ ... GCNNNNN^NNGC ... 3´ 3’ overhang 3´ ... CGNN^NNNNNCG ... 5´ (3 bp)
How often does REase cut my sequence? • Known sequence: scan for sites by computer (eg. at www.rebase.neb.com) • Unknown sequence: hypothetical calculations 4 cutter: site occurs randomly every 44 (256) base pairs 6 cutter: every 46 (4096) bp 8 cutter: every 48 (65536) bp But sequences are not distributed randomly (table 3.4) • Sequence context effects Some sites are preferred over others by enzyme
The ligation reaction • Biological function of ligases: • Lagging strand DNA synthesis • genetic recombination • DNA repair
Cloning techniques A) Modify the ends of the DNAs to make foreign DNA sequences more ligate-able B) Directional cloning (generate easily cloned PCR fragments) C) Treat the vector DNA with alkaline phosphatase to improve the efficiency of ligation of foreign DNA versus vector recircularization
Creating a recombinant DNA molecule Plasmid vector: a cloning vehicle it can replicate itself in a bacterial host and contains a means for selection (eg. antibiotic resistance)
Ligation efficiency depends on the DNA ends in the reaction • Complementary “sticky” ends • Ligation is efficient • annealing of complementary overhangs brings 5’P and 3’OH into close proximity • “Blunt” ends • Ligation is inefficient • need high concentrations of ligase and DNA • molecular crowding reagents (like PEG 8000) improve intermolecular ligation, then dilute to promote intramolecular ligation • Follow the manufacturer’s instructions…
Cloning foreign DNA by adding linkers (your DNA molecule should not have EcoRI sites in this case)
Cloning foreign DNA by adding adaptors The advantage of this is you do not need to treat the adaptor-modified DNA with restriction enzyme
Terminal transferase to add polynucleotide tails to foreign DNA and vector DNA Foreign DNA Vector DNA dTTP
Cloning Taq PCR products • Taq PCR products have a 3’ “A” overhang • Prepare vector to have a 3’ “T” overhang HphI leaves T overhangs
Directional cloning This guarantees the orientation of your DNA fragment
Easy cloning: PCR products Design PCR primers with built in restriction sites (check amplified sequence for those sites first!) Ready for directional cloning
Utility of alkaline phosphatase in ligation Chances of getting recombinant product are improved
Cutting and pasting DNA • Restriction and modification systems • Recognition and cleavage of DNA by restriction endonucleases (REases) • Joining (ligating) DNA molecules • Cloning techniques