Thermochemical Analysis of Magnesium and Oxygen Reaction to Form Magnesium Oxide
10 likes | 181 Vues
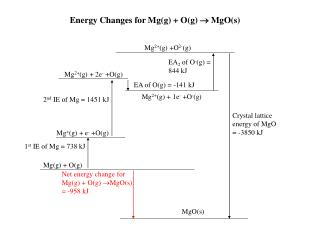
This document analyzes the energy changes associated with the formation of magnesium oxide (MgO) from magnesium (Mg) and oxygen (O) gases. The reaction can be represented as Mg(g) + O(g) → MgO(s), with a net energy change of -958 kJ. The enthalpy of formation includes the electron affinity (EA) of oxygen gases at 844 kJ, the first ionization energy (IE) of magnesium at 738 kJ, and the crystal lattice energy of magnesium oxide at -3850 kJ, highlighting the exothermic nature of this process.

Thermochemical Analysis of Magnesium and Oxygen Reaction to Form Magnesium Oxide
E N D
Presentation Transcript
Energy Changes for Mg(g) + O(g) MgO(s) Mg2+(g) +O2-(g) EA2 of O-(g) = 844 kJ Mg2+(g) + 2e- +O(g) EA of O(g) = -141 kJ Mg2+(g) + 1e- +O-(g) 2nd IE of Mg = 1451 kJ Crystal lattice energy of MgO = -3850 kJ Mg+(g) + e- +O(g) 1st IE of Mg = 738 kJ Mg(g) + O(g) Net energy change for Mg(g) + O(g) MgO(s) = -958 kJ MgO(s)