pH Scale and Calculations
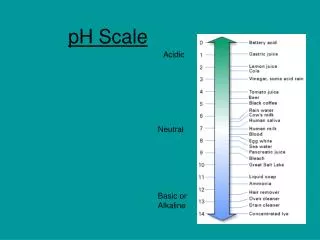
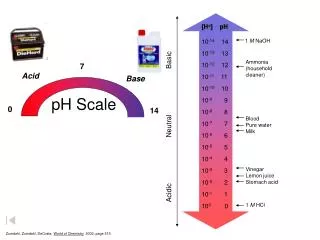
pH Scale and Calculations. Chapter 14 Page 565-574. pH Scale . We use this scale to measure the strength of an acid or base. pH is defined as the –log[H+] pH can use the concentration of hydronium ions or hydrogen ions. pH Scale. 7. Acid. Base. 0. 14.

pH Scale and Calculations
E N D
Presentation Transcript
pH Scale and Calculations Chapter 14 Page 565-574
pH Scale • We use this scale to measure the strength of an acid or base. • pH is defined as the –log[H+] • pH can use the concentration of hydronium ions or hydrogen ions.
pH Scale 7 Acid Base 0 14 Zumdahl, Zumdahl, DeCoste, World of Chemistry2002, page 515
pH of Common Substances Timberlake, Chemistry 7th Edition, page 335
pH of Common Substance pH [H1+] [OH1-] pOH 14 1 x 10-14 1 x 10-0 0 13 1 x 10-13 1 x 10-1 1 12 1 x 10-12 1 x 10-2 2 11 1 x 10-11 1 x 10-3 3 10 1 x 10-10 1 x 10-4 4 9 1 x 10-9 1 x 10-5 5 8 1 x 10-8 1 x 10-6 6 6 1 x 10-6 1 x 10-8 8 5 1 x 10-5 1 x 10-9 9 4 1 x 10-4 1 x 10-10 10 3 1 x 10-3 1 x 10-11 11 2 1 x 10-2 1 x 10-12 12 1 1 x 10-1 1 x 10-13 13 0 1 x 100 1 x 10-14 14 NaOH, 0.1 M Household bleach Household ammonia Lime water Milk of magnesia Borax Baking soda Egg white, seawater Human blood, tears Milk Saliva Rain Black coffee Banana Tomatoes Wine Cola, vinegar Lemon juice Gastric juice More basic 7 1 x 10-7 1 x 10-7 7 More acidic
Acid – Base Concentrations 10-1 pH = 3 pH = 11 OH- H3O+ pH = 7 10-7 concentration (moles/L) H3O+ OH- OH- H3O+ 10-14 [H3O+]<[OH-] [H3O+]>[OH-] [H3O+]=[OH-] acidic solution neutral solution basic solution Timberlake, Chemistry 7th Edition, page 332
pH pH = -log [H+] Kelter, Carr, Scott, Chemistry A World of Choices 1999, page 285
Self-Ionization Of Water • Even the purest of water conducts electricity. This is due to the fact that water self-ionizes, that is, it creates a small amount of H3O+ and OH-. H2O + H2O º H3O+ + OH- Kw = [H3O+][OH-] • Kw - ion product of water Kw = 1.0 x 10-14 at 25 oC • This equilibrium constant is very important because it applies to all aqueous solutions - acids, bases, salts, and non-electrolytes - not just to pure water.
O H H H Self ionization reaction of water: + - O O + O + H H H H H
pH and pOH • pH = - log[H3O+] [H3O+] = 10-pH pOH = - log[OH-] [OH-] = 10-pOH • pKw = pH + pOH = 14.00 • neutral solution: [H3O+] = [OH-] = 10 –7 M pH = 7.0 acidic solution: [H3O+] > 10-7 M pH < 7.0 basic solution: [H3O+] < 10-7 M pH > 7.0
Practice • Finish the following for homework Page 566 #12-15 (using Kw) Page 569 # 16-19 (using pH = -log[H+] Page 572 # 20-23 Page 578 # 2, 6-8