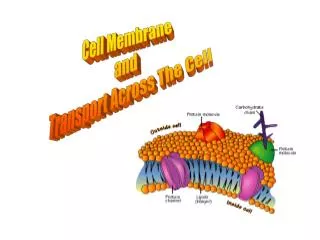
Cell Membrane/Plasma Membrane
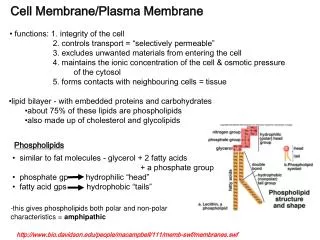
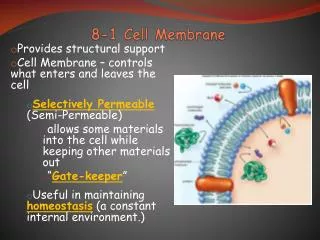
similar to fat molecules - glycerol + 2 fatty acids + a phosphate group phosphate gp hydrophilic “head” fatty acid gps hydrophobic “tails”. Cell Membrane/Plasma Membrane. functions: 1. integrity of the cell 2. controls transport = “selectively permeable”


Cell Membrane/Plasma Membrane
E N D
Presentation Transcript
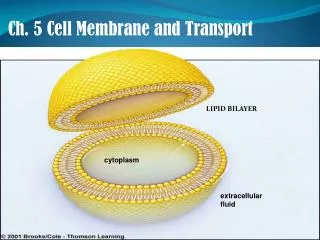
similar to fat molecules - glycerol + 2 fatty acids • + a phosphate group • phosphate gp hydrophilic “head” • fatty acid gps hydrophobic “tails” Cell Membrane/Plasma Membrane • functions: 1. integrity of the cell • 2. controls transport = “selectively permeable” • 3. excludes unwanted materials from entering the cell • 4. maintains the ionic concentration of the cell & osmotic pressure • of the cytosol • 5. forms contacts with neighbouring cells = tissue • lipid bilayer - with embedded proteins and carbohydrates • about 75% of these lipids are phospholipids • also made up of cholesterol and glycolipids Phospholipids -this gives phospholipids both polar and non-polar characteristics = amphipathic http://www.bio.davidson.edu/people/macampbell/111/memb-swf/membranes.swf
A. Composition: polar heads out non-polar tails in -the polar and non-polar attributes of the lipids results in a bilayer arrangement -cholesterol is also polar (OH group) and non-polar (steroid rings) and contributes to this arrangement – OH group faces out and the steroid rings face inward
1. peripheral or extrinsic -bind to the outside only e.g. enzymes 2. integral or intrinsic -globular and amphipathic -can span 1 or both layers -most are transmembrane(long, rodlike) • many lipids are proteins are modified by the attachment • of carbohydrates = ‘glyco’proteins & ‘glyco’lipids • glycoproteins & glycolipids form a superficial coat around the • cell = ‘glycocalyx’ • membrane proteins
Functions of Integral Proteins -in addition: 4. enzymes 5. linkers – anchor proteins of the PM to the protein filaments inside or to neighboring cells 6. cell-identity markers – used in identifying “self” by the immune system e.g MHC proteins e.g. ABO blood typing
ion channels = gates for specific ions only • -open in response to: 1. changes in voltage • 2. binding of a ligand • e.g. calcium • sodium • chloride • potassium • -affected by drugs • e.g. anti-hypertensives - calcium, potassium • local anesthetics - sodium • diuretics - sodium • muscle relaxants - chloride • anti-diabetics - potassium • -disease states affect channel function • e.g. cystic fibrosis
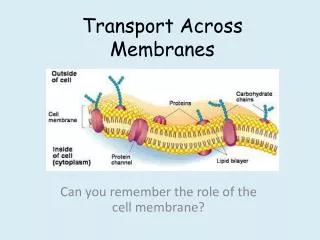
B. Membrane function: • Physical isolation - from the surrounding ECF • -allows the cell to create different environments outside and inside • -allows for the creation of gradients – electrical and chemical 2. Integrity of cell - cell shape and size -increase cell size, increase surface area/volume -increase exchange surface 3. Sensitivity - first part of cell that is affected by changes in the extracellular environment 4. Structural support - connections between cells provides tissues with support and stability 5. Controls transport = “selectively permeable” -two types: Passive - Diffusion, Osmosis, Facilitated Active - Active transport, Exocytosis, Endocytosis,
Membrane Gradients • selective permeability of the PM allows the cells to control the concentration of ions within the cell and outside the cell (in the ECF) • this results in a distinct distribution of positive and negative ions inside and outside the cell • typically the inside of the cell is more negatively charged • this difference in electrical charge between inside and outside = electrical gradient • because it occurs across the PM – we call this difference in charge = membrane potential • can be measured with tiny glass electrodes • varies from cell to cell • very important in the functioning of neurons and muscle cells
Membrane Permeability and Transport • permeability = property that determines the effectiveness of the PM as a • barrier • permeability varies depending on the organization and characterization of • the membrane lipids and proteins • transport across the membrane may be passive or active passive transport active transport diffusion osmosis facilitated endocytosis (pinocytosis phagocytosis receptor-mediated) exocytosis http://programs.northlandcollege.edu/biology/Biology1111/animations/transport1.html
materials may cross into a cell based on concentration and size • if they cross from [high] to [low] – they are traveling with their concentration • gradient – requires no energy (Passive) • -if they cross against the concentration gradient – requires energy (Active) • small particles may cross through the lipid bilayer • others may require integral proteins that help (e.g. channels or pores) • others may enter through the fusion of tiny vesicles with the PM
A. Diffusion = movement of materials from [high] to [low] -random movement, no energy needs to be synthesized -the movement is driven by the inherent kinetic energy of the particles moving down their concentration gradient -movement could be through the bilayer itself or through channel proteins -three ways to diffuse: 1. through the lipid bilayer: lipid soluble (non-polar), alcohol, gases, ammonia, fat-soluble vitamins 2. through a channel: charged, small ions (polar) -some channels are “gated” – open and close 3. facilitated diffusion: larger molecules too big for channels
B. Osmosis = diffusion of water from [high] to [low] OR movement of water from [low solute] to [high solute] -in osmosis – the membrane is permeable to water and NOT to the solutes -but it is the concentration of solutes that causes the water to move -experiment – U shaped tube divided by a membrane permeable to water only -increase the solute concentration in the right half of the tube -this increases the pressure caused by the increase solutes = osmotic pressure -therefore increasing solute concentration increases osmotic pressure -water will move in to decrease this OP -OP is important in determining how much fluid remains in your blood and how much leaves to surround the cells in your tissues
hypertonic hypotonic = [S]in > [S]out, water enters cell -Osmosis is controlled by tonicity = degree to which a the concentration of a specific solute surrounding a cell causes water to enter or leave the cell hypertonic = [S]in < [S]out, water exits cell e.g. isotonic = [S]in = [S]out, no water movement -medical uses of solutions requires careful consideration of osmolarity e.g. can cause destruction of red blood cells if these cells are placed in hypotonic or hypertonic solutions -typical saline solutions are 0.9% NaCl = isotonic saline -other IV solutions are also isotonic e.g. D5W – 5% dextrose in water -but hypertonic and hypotonic solutions can be used in specific situations e.g. cerebral edema = water is forced out of the blood and into the brain tissue -treatment with hypertonic saline causes water to leave the brain tissue back into the where it is removed by the kidneys e.g. dehydration – treatment with hypotonic solutions to increase water content of ECF
C. Facilitated transport = molecules move by a carrier protein from [high] to [low] -binds to a receptor site on the plasma membrane -transported by the carrier protein -no energy required -but there is a limit to the amount of FD cells can undergo and it has to do with the # of carrier proteins on the PM -molecules that are insoluble, too polar or too large e.g. glucose amino acids
Medical application -the number of transporters during homeostasis remains constant -but cells can increase or decrease the expression of these carriers in response to the environment -increased blood sugar – production of insulin by the pancreas - insulin causes cells (e.g. adipose cells, liver cells, muscle cells) to increase their expression of a glucose transporter (GLUT proteins) on the surface -this increases the uptake of sugar from the blood -failure to produce enough insulin or failure of cells to express GLUT transporters in response to insulin = diabetes mellitus
A. Active transport = molecules are moved against the the concentration gradient i.e. from [low] to [high] -two kinds: primary and secondary -primary active transport: -requires a protein carrier and ATP -carrier is often called a pump -ATP binds to the pump and changes its shape (ATPase) e.g. sodium/potassium pump – three Na are pumped out of a cell and 2 K are pumped into the cell (Na/K ATPase) -maintains a specific concentration of Na within the cell and K outside the cell -Na binds to the pump, ATP then binds and hydrolyzes, a P group attaches to the pump and changes its shape – expels the Na out of the cell -K then binds the pump and causes the release of the P, the pump returns to its original shape, bringing K into the cell http://highered.mcgraw-hill.com/sites/0072437316/student_view0/chapter6/animations.html#
2. secondary active transport: -the energy stored in a concentration gradient is used to drive the transport of other materials e.g Na/Ca antiporter – opposite direction for Na and Ca movement – primary transport establishes high [Na] outside the cell – this concentration gradient creates potential energy which is stored by the antiporter pump - as Na leaks back in – this potential energy is converted into kinetic energy which drive the movement of a Ca ion against its gradient -some pumps can also pump two materials in the same direction = symporter e.g. Na/glucose symporter -most of our cells use the energy created by the Na gradient to power the movement of other ions diffusion diffusion Na pump diffusion diffusion http://highered.mcgraw-hill.com/sites/0072437316/student_view0/chapter6/animations.html# Low Na, low Ca High glucose, high amino acids
-primary active transport and ATP hydrolysis pump Na out of the cell and creates a sodium gradient -increased sodium gradient = increased membrane potential energy -when sodium diffuses back into the cell through the symporter or antiporter, potential energy is converted into kinetic energy and the second ion can be pumped against its gradient -same direction as Na = symporter -opposite direction as Na = antiporter
B. Exocytosis = secretion of a substance outside the cell -made within the cell, packaged into transport vesicles-> fusion with the plasma membrane and release outside the cell e.g. nerve cells - neurotransmitter release http://highered.mcgraw-hill.com/sites/0072437316/student_view0/chapter6/animations.html#
C. Endocytosis = reverse of exocytosis, internalization of substances -3 forms: 1. pinocytosis = “cell drinking”
3. receptor-mediated = internalization of specific substances -binding of a ligand with its receptor -> internalization into the cell -occurs at specific sites within the PM -> clathrin-coated pits -internalization at pits -> clathrin-coated vesicle -vesicle fuses with endosomes - processing
Medical application • HIV and receptor-mediated endocytosis • binding of HIV virus to the CD4 protein on the surface of T helper cells and macrophages results in the RME of the HIV virus • the HIV viral particles are made by the host cell protein synthesis machinery and assembled at the host’s PM – released from the cell = exocytosis • the infected T cells are killed leading to low T cell counts in infected people