Physical Science
Physical Science. Final Exam Review. Physical Science Final Exam Review Game Board. What is the difference between a chemical and physical property? Give an example of each. . Game Board. What is the difference between a physical and chemical change. Give an example of each. Game Board.

Physical Science
E N D
Presentation Transcript
Physical Science Final Exam Review
Physical Science Final Exam Review Game Board
What is the difference between a chemical and physical property? Give an example of each. Game Board
What is the difference between a physical and chemical change. Give an example of each. Game Board
What are the three states of matter? Describe 3 characteristics of each one. Game Board
What is the difference between a pure substance and a mixture. Give an example of each. Game Board
What is the difference between an element, compound, and a mixture? Give an example of each. Game Board
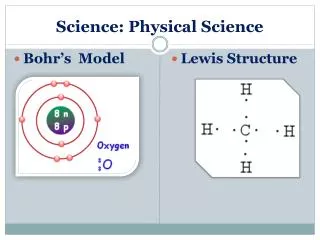
What is the modern definition of the term “atom?” Game Board
What is the difference between a heterogeneous and a homogeneous mixture? Give an example of each. Game Board
Which state of matter has the greatest amount of potential energy? Game Board
What is the process by which a solid changes directly to a gas? Game Board
Fill in the Blank: Changes in state occur at ________ temperatures. Game Board
What are the two types of vaporization. Explain how they are different. Game Board
What is “heat of fusion?” Game Board
What is matter? Game Board
What two types of substances are considered “pure substances?” Game Board
Who thought that matter was “infinitely divisible?” Game Board
Please list the number of protons, neutrons, and electrons for the following ion… Game Board
Please list 5 physical properties that can be measured of metals. Game Board
What does it mean if a metal is “ductile?” Game Board
Mendeleev arranged the periodic table by increasing _________. Game Board
Dobereiner’s method of arranging elements was called _______. Game Board
Who is the scientist who discovered a repeating pattern in properties every eight elements? Game Board
Which scientist left blank spaces for elements that had not been discovered yet in the periodic table? Game Board
Describe the location, size, and charge on an electron. Game Board
What do elements in the same period in the periodic table have in common? Game Board
Indicate what charge a magnesium atom will acquire when it becomes an ion and WHY. Game Board
Which group of metals are the most reactive group on the periodic table? Game Board
What is meant by the word “inert” and which group of elements can it be used to describe? Game Board
What do elements in the same group in the periodic table have in common? Game Board
What is the octet rule? Game Board
Describe the role of valence electrons in ionic and covalent bonds. Game Board
What physical property can be observed in ionic compounds but NOT molecular compounds when dissolved in water? Game Board
List the 10 prefixes used in naming molecular compounds. Game Board
Formulas for ionic compounds are always _________. Game Board
Name this compound: MgCl2 Game Board
Name this compound: NO2 Game Board
Name this compound: P2O4 Game Board
Name this compound: Ca(OH)2 Game Board
Write the formula for this compound: Dinitrogen Pentoxide Game Board
Write the formula for this compound: Iron (III) Carbonate Game Board
Write the formula for this compound: Sodium Nitride Game Board
Write the formula for this compound: Carbon Tetrachloride Game Board
Write the formula for this compound: Ammonium Sulfide Game Board
List the 5 pieces of evidence that support a the occurrence of a chemical reaction. Game Board
Write and balance the equation for this reaction: Iron reacts with copper (II) sulfate to produce copper and iron (II) sulfate. Game Board
Write and balance the equation for this reaction:Lead (II) nitrate and aluminum chloride react to produce lead (II) chloride and aluminum nitrate. Game Board
Write and balance the equation for this reaction:Zinc and hydrogen chloride react to produce zinc chloride and hydrogen. Game Board
Write and balance the equation for this reaction:Hydrogen and oxygen react to produce water. Game Board