Acid-Base Buffers
© Mr. D. Scott; CHS. Conjugate Base. Weak Acid. Acid-Base Buffers. A BUFFER is a mixture whose pH changes very little when a small amount of strong acid or base is added to it.


Acid-Base Buffers
E N D
Presentation Transcript
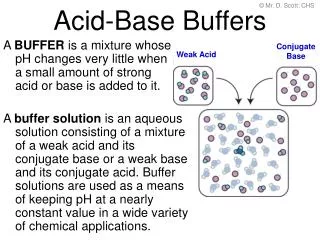
© Mr. D. Scott; CHS Conjugate Base Weak Acid Acid-Base Buffers A BUFFER is a mixture whose pH changes very little when a small amount of strong acid or base is added to it. A buffer solution is an aqueous solution consisting of a mixture of a weak acid and its conjugate base or a weak base and its conjugate acid. Buffer solutions are used as a means of keeping pH at a nearly constant value in a wide variety of chemical applications.
© Mr. D. Scott; CHS Acid-Base Buffers Buffers in the Human Body
© Mr. D. Scott; CHS Acid-Base Buffers pH and H+ concentration of body fluids Since the normal pH of the arterial blood is 7.4, a person is considered to have acidosis whenever the pH is below this value and to have alkalosis when it rises above 7.4. The lower limit at which a person can live more than a few hours is about 6.8, and the upper limit approximately 8.0.Defenses against changes in hydrogen ion concentration buffers, lungs and kidneys
© Mr. D. Scott; CHS Acid-Base Buffers
© Mr. D. Scott; CHS Acid-Base Buffers There are three primary systems that regulate the hydrogen ion concentration in the body fluids. • The chemical acid base buffer system of the body fluids, which immediately combine with acid or base to prevent excessive changes in hydrogen ion concentration. • The respiratory center which regulates the removal of CO2 (and therefore H2CO3) from the extracelluar fluid. • The kidneys which can excrete either acid or alkaline urine, there by readjusting the extra cellular fluid hydrogen ion concentration toward normal during acidosis or alkalosis.