Effective Enrollment Strategies in Research Management
130 likes | 228 Vues
This presentation covers key enrollment strategies in research management, including regulatory compliance, subject eligibility, and study protocols. Learn how to navigate enrollment challenges effectively.

Effective Enrollment Strategies in Research Management
E N D
Presentation Transcript
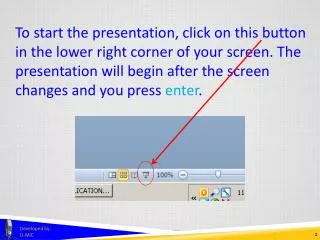
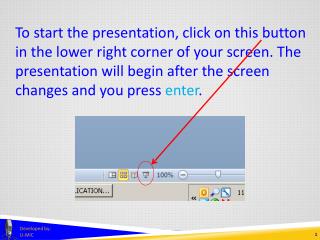
To start the presentation, click on this button in the lower right corner of your screen. The presentation will begin after the screen changes and you press enter. Developed by: U-MIC
Human subjects enrollmentin eResearch Developed by: U-MIC University of Michigan IRB Collaborative
maximum number of subjects to be enrolled Enrollment eResearch Regulatory ManagementSection 08 * * Section 03-2 for Cancer Center studies Developed by: U-MIC
scheduled continuing review (SCR) application Enrollment eResearch Regulatory ManagementSection 02-2 (SCR) Developed by: U-MIC
over-enrollment • violation of IRB-approved protocol • particularly concerning with research that presents more than minimal risk • under-enrollment • may result in insufficient data Enrollment Developed by: U-MIC
U-M standard definition of enrolled Enrollment consented and screened,with eligibility verified • main application: section 08 (section 03-2 for Cancer Center studies) • SCR application: section 02-2 • subjects enrolled = number of subjects expectedto complete the study • includes dropouts (withdrawals) • does not include screen failures Developed by: U-MIC
enrollment • applies to studies involving intervention or interaction with individual subjects • does not apply to secondary use of samples and/or data Enrollment Developed by: U-MIC
inclusion and exclusion criteria • age / other demographics • health • characteristics • screening • may or may not involve interaction or intervention • may take place before informed consent(waiver of consent for recruitment) • may take place after informed consent Enrollment Developed by: U-MIC
At SCR, report individuals determined ineligible as screen failures in eResearch section 02-2.2. Enrollment Developed by: U-MIC
protocol-specific definitions of enrolled • common in multi-site trials with outside sponsorship • may differ from U-M’s standard definition Enrollment Developed by: U-MIC
MBECT(Michigan Budget Enrollment Calendar Tool) • clinical trials • separate from eResearch • MBECT subject enrollment record for each consented participant • Contact CRAO (the Calendar Review and Analysis Office) for clinical research billing help. Enrollment Developed by: U-MIC
Enrollment • eResearch Release Notes Version 3.2 • http://www.umich.edu/~eresinfo/errm/rnotes/releasenote_v3.2.html • Enrolledfor human subjects studies in eResearch • http://medicine.umich.edu/medschool/sites/medicine.umich.edu.medschool/files/res_irbmed_EnrollmentDefinition_AdditionalHelp.pdf • CRAO • http://medicine.umich.edu/medschool/research/office-research/calendar-review-analysis-office/mbect Developed by: U-MIC
Lark Speyer IRBMED thank you. Developed by: U-MIC