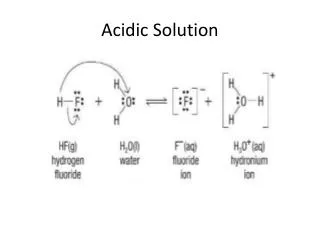
Acidic redox
E N D
Presentation Transcript
RA’s Balancing acidic redox Questions 3,4 and 5
Q3)P + SO42- SO2+ PO43- 2 x (4H2O + P PO43- + 8H+ + 5e-) 5x ( 2e- + 4H+ + SO42- SO2 + 2H2O) 8H2O +2P 2PO43- + 16H+ + 10e- 10e-+ 20H+ +5SO42- 5SO2 + 10H2O 2P + 4H+ +5SO42- 2PO43- + 5SO2 + 2H2O
Q4) I- + SO42- I2 + SO2 2I- I2 + 2e-½ oxidation reaction 2e- + 4H+ + SO42- SO2 + 2H2O ½ reduction reaction 2I- + 4H+ + SO42- I2 + SO2 + 2H2O complete redox reaction Which reactant is reducing agent?
Q5) I- + SO42- I2+ SO32- 2I- I2 + 2e- 2e- + 2H+ + SO42- SO32- + H2O 2I- + 2H+ + SO42- I2 + SO32- + H2O What is oxidation number of sulfur in SO42- & SO32- ? What specie/element has been reduced? Witch reactant is oxidizing agent?