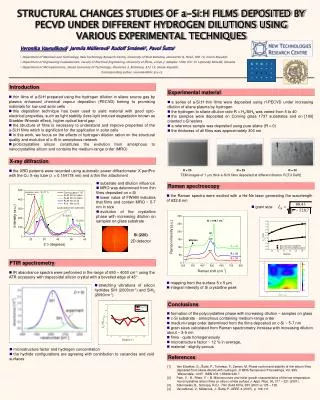
Dilutions!
Dilutions!. Mix 2 solutions together…. …they both get diluted. It’s ping pong balls in a bucket…. 5 gallon bucket - Ping pong ball. 5 gallon empty bucket - 5 Ping pong ball. Add 1 gallon of water. What’s the “concentration of ping pong balls”? 5 PPB/gallon!. Add another gallon of water.

Dilutions!
E N D
Presentation Transcript
Mix 2 solutions together… …they both get diluted. It’s ping pong balls in a bucket…
Add 1 gallon of water What’s the “concentration of ping pong balls”? 5 PPB/gallon!
Add another gallon of water What’s the “concentration of ping pong balls”? The ping pong balls are no different than atoms or molecules. If you have 5 moles in a bucket, you have 5 moles in a bucket!
5 gallon empty bucket Add 5 ping pong balls and 2 baseballs.
Add 1 gallon of water What’s the “concentration of ping pong balls”? 5 PPB/gallon! What’s the concentration of baseballs? 5 BB/gallon
Add another gallon of water What’s the “concentration of ping pong balls”? The concentration of base balls is:
If they are different molecules… The concentration gets calculated separately!
Add 1 gallon of water 2 BB in 1 gallon of water! 2 BB/gal
Add 2 gallon of water 5 PPB in 2 gallons of water 2.5 PPB/gal
Suppose I mix the two bukets 2 gallons + 1 gallon = 3 total gallons. The number of objects is unchanged.
And what if the objects are the same? They just add together!
Add 2 gallon of water 5 PPB in 2 gallons of water 2.5 PPB/gal
Add 2 gallon of water 3 PPB in 2 gallons of water 1.5 PPB/gal
Suppose I mix the two bukets 2 gallons + 1 gallon = 3 total gallons of “solvent”. The number of ping pong balls also adds: 5 PPB + 3 PPB = 8 total PPB
Same with chemical solutions… EXCEPT it’s usually Liters (or mL) of solvent… And it is MOLES! MOLES! MOLES! of solute
I mix 250 mL of 0.250 M NaCl with 500 mL of 0.100 M HCl. What is the concentration of NaCl? 0.250 M x 0.250 L = 0.0625 molNaCl The problem doesn’t ask about HCl, but it is the same issue – dilution: 0.100 M x 0.500 L = 0.05 molHCl
I mix 250 mL of 0.250 M NaCl with 500 mL of 0.100 M NaCl. What is the concentration of NaCl? 0.250 M x 0.250 L = 0.0625 molNaCl 0.100 M x 0.500 L = 0.05 molNaCl