Alkenes
16/07/2014. Alkenes. L.O. I can draw the structural and molecular formula of the first four alkenes. I know the test for alkenes and can use it to identify an alkene from an alkane. C. C. Single covalent bond. C. C. Double covalent bond. Alkenes.

Alkenes
E N D
Presentation Transcript
16/07/2014 Alkenes L.O. • I can draw the structural and molecular formula of the first four alkenes. • I know the test for alkenes and can use it to identify an alkene from an alkane.
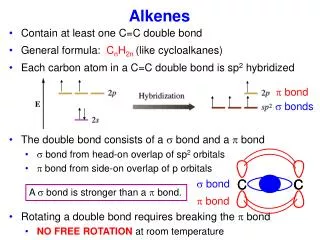
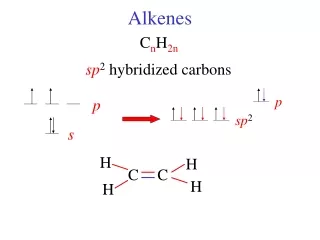
C C Single covalent bond C C Double covalent bond Alkenes • When carbon forms compounds each carbon atom always forms four bonds. • This does not, however, mean that each carbon is joined to four other atoms. • It is possible to have bonds grouped into pairs. These are called double bonds. • Alkenes contain carbon atoms joined by double covalent bonds.
H H C C H H or Ethene - formula • The simplest alkene is ethene. • It has the formula C2H4 • The carbon atoms are joined together by a double bond. • Its displayed formula may be drawn in slightly different forms but should always clearly show the double bond.
16/07/2014 Alkenes Molecular formula Displayed formula Ethene C2H4 HH C C HH Propene C3H6 HH H C C C H HH
16/07/2014 Alkenes Molecular formula Displayed formula Butene C4H8 HH H H C C C C H HH H Pentene C5H10 HH H H H C C C C C H HH HH What is the general formula for the alkenes? – CnH????
Activity H H H C H C C H H H H H H C H H C H H C H C H H H C C C H H C C H H H H • Sometimes the displayed formula is drawn as shown.
Activity • What will be the formula for alkenes containing the following numbers of carbons? C11H22 C13H26 C32H64 C21H42
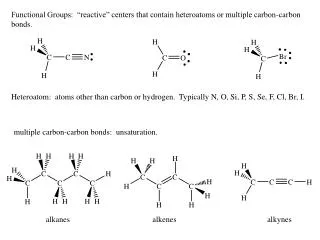
Saturated or Unsaturated? • Alkanes are saturated. • Saturated means “full up”. • Every carbon atom has already used all four of it’s bonds to join to four other atoms. No other atoms can be added. • Alkenes are unsaturated. • They have a double bond that could instead become two single bonds. This means that other atoms can be added. It is not “full up”.
Br2 + Gas red colourless Bromine water +alkene Test for Alkenes When bromine water is added to an alkane nothing happens but when bromine is added to an alkene the red colour of the bromine disappears. 1,2 dibromoethane
Activity • Complete the blanks alkene C5H10 alkane C8H18 alkene C4H8 alkane C10H22
Which of these is an alkene? • C6H14 • C4H8 • C12H26 • C102H206
The formula of the alkene with 15 carbons is? • C15H30 • C15H60 • C15H32 • C15H28
Hydrocarbons contain only which two elements? • Carbon and electron • Carbon and water • Carbon and oxygen • Carbon and hydrogen
The bonds in a hydrocarbon are what type? • Covalent • Ionic • Complex • Metallic
Which is the simplest Alkene? • Methene • Decene • Ethene • Butene
Which is alkene has 5 carbons? • Propene • Pentene • Pectane • Proptene
What is the molecular formula of Butene? • C3H6 • C4H8 • C2H4 • C6H12
What do alkanes do to Bromine water? • Produce layers • Turn it colourless • Leave it unchanged
Bingo 1. Alkanes 9. Bromine water 2. Methane 10. covalent 3. C2H6 11. propane 4. Carbon 12. colourless 5. Hydrogen 13. electron 6. Orange 14. C2H4 7. ethene 15. alkene 8. Six 16. butene
Homework Complete sheet on the alkenes.