Isotopes
0. Isowhat? Man, you trippin’ nephew. Isotopes. 0. 0. 0. What Are Isotopes ?. They are atoms of the same element that have different numbers of neutrons. It’s just a change in the neutrizzle fo shizzle!!!. 0. An Example. Hydrogen has 3 isotopes

Isotopes
E N D
Presentation Transcript
0 Isowhat? Man, you trippin’ nephew. Isotopes
0 What Are Isotopes? • They are atoms of the same element that have different numbers of neutrons. It’s just a change in the neutrizzle fo shizzle!!!
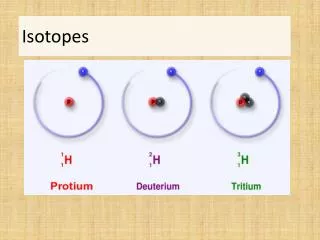
0 An Example • Hydrogen has 3 isotopes • There are 3 different “versions” of a hydrogen atom • 1 proton and 0 neutrons (mass number = 1) • 1 proton and 1 neutron (mass number = 2) • 1 proton and 2 neutrons (mass number = 3) Note how the number of protons never change!!!
0 Ever Wonder Why The Massesof Atoms Are Such Weird Numbers? • For example, carbon’s mass is 12.01115 amu • amu means atomic mass unit • amu’s are a unit created specifically for measuring the extremely small masses of atoms • Another example, neon’s mass is 20.183 amu
0 Why All the Decimal Places? • Atomic mass is the “weighted” average of all the different isotopes of that element.
0 Example: Magnesium • Has 3 isotopes
0 % abundance?????? • Every isotope is found in nature in a fixed percentage. • In other words, 79% of the magnesium atoms in any “scoop” of magnesium found anywhere in the universe will be magnesium atoms with 12 neutrons, 10% of the atoms in the scoop will have 13 neutrons, and 11% will have 14 neutrons. • These percentages are different for each element…you just have to look them up to find out what they are.
0 Calculating Atomic Masses • For magnesium, the atomic mass is given by… 23.985045(.79) +24.985839(.10) +25.982595(.11) 24.30486 amu’s Since its a “weighted” average this number has more influence since this mass occurs 79% of the time It’s NOT just a straight average where you add them up and divide by 3 Note how the % abundances were converted to decimals (just divide each by 100)
0 The General Formula for Calculating Atomic Masses Is… (mass of isotope 1)(% abundance/100) +(mass of isotope 2)(% abundance/100) +(mass of isotope 3)(% abundance/100) +’keep going if there are more isotopes’
0 O.K. nephew…try these examples, then we can chill ‘till the next episode…
0 Calculate the Atomic Massof Carbon
0 For Carbon… 12.000000(98.90 / 100) + 13.003355(1.10 / 100)
0 Calculate the Atomic Massof Chromium
0 For Chromium… 49.946046(4.35/100) 51.940510(83.79/100) + 52.940651(9.50/100) 53.938882(2.36/100)
0 The Vocabulary Section Terms associated with isotopes
0 A Few Important TermsYa Gotta Know • Atomic number = the number of protons • It’s the same as the element’s position on the periodic table • For example, carbon is the 6th element on the table, so its atomic number is 6 which means it has 6 protons (and 6 electrons if it’s a neutral atom)
0 A Few Important TermsYou Gotta Know • Atomic mass = the “weighted” average mass of all the different isotopes of an atom • This is the “weird” number on the periodic table that usually has a bunch of places past the decimal point
0 A Few Important TermsYou Gotta Know • Mass number = the sum of protons + neutrons • This one kind of makes sense because remember we said the electrons essentially don’t have any mass • This HAS to be a wholenumber!!!!! • It has to be, because you can’t have part of a proton or neutron • Mass number is NOT given on the periodic table
0 Answers • 12 and 13 • 7 • 19 • 10
0 X Notation for Isotopes A Z A shorthand way to write isotopes
0 A Short Way to Write Isotopes “A” is the symbol for Mass Number (#protons + #neutrons) X A Z “Z” is the symbol for Atomic Number (# of protons) “X” will be the symbol of the element
Jay - Z Yo! If you’re doin’ chem problems I feel bad for you son…. I got 99 problems, and here’s the first one….HIT ME!!!!
Problem #1 • Write az X notation for an isotope that has 17 protons and 18 neutrons.
Problem #2 • Write azX notation for an isotope that has 19 protons and 21 neutrons.
Problem #3 • Write azX notation for an isotope that has 15 protons, 18 electrons, and 16 neutrons.
Answer: P3- This isotope is also an ion!!! 31 15
Problem #4 • How many protons, electrons, and neutrons are in this isotope? • 20Ca2+ 41
Problem #5 • How many protons, electrons, and neutrons are in: • 56Fe2+ • 27Al3+ • 79Se2- 26 13 34
Problem #5 Answers • 26, 24, 30 • 13, 10, 14 • 34, 36, 45
Problem #6 Write the proper notation for: • Isotope with 21 protons, 18 electrons, and 24 neutrons • Isotope with 53 protons, 74 neutrons, and 54 electrons
Problem #6 Answers • 45Sc3+ • 127I- 21 53
Different Forms of AX Z • 3517Cl is the same as 35Cl17 • Might also see “Cl-35” or “chlorine-35”