PHYSICAL MAPPING AND POSITIONAL CLONING
PHYSICAL MAPPING AND POSITIONAL CLONING. Linkage mapping – Flanking markers identified – 1cM, for example Probably ~ 1 MB or more in humans Need very many families to get closer than this in human, or very large populations. What to Do Next?. Identify genes in this region

PHYSICAL MAPPING AND POSITIONAL CLONING
E N D
Presentation Transcript
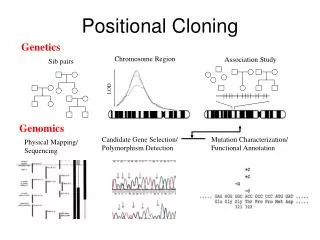
Linkage mapping – Flanking markers identified – 1cM, for example • Probably ~ 1 MB or more in humans • Need very many families to get closer than this in human, or very large populations
What to Do Next? • Identify genes in this region • Then determine what is the gene of interest
What to do next? • In 1990, clone region around markers, make physical map(s), look for genes experimentally • In 2000, use better physical maps, at least in some organisms • In 2010, use bioinformatic knowledge, experimental proof still necessary
Physical Mapping A physical map is primarily based on the locations of landmarks along a DNA molecule and units of distance are expressed in base pairs.
Low Resolution Physical Mapping • Cytogenetic map • In situ hybridization
Chromosome Sorting • Flow cytometry • Used in library construction • Also for chromosome paints
Polymerase Chain Reaction (PCR) • Ability to generate identical high copy number DNAs made possible in the 1970s by recombinant DNA technology (i.e., cloning). • Cloning DNA is time consuming and expensive (>>$15/sample). • Probing libraries can be like hunting for a needle in a haystack. • PCR, “discovered” in 1983 by Kary Mullis, enables the amplification (or duplication) of millions of copies of any DNA sequence with known flanking sequences. • Requires only simple, inexpensive ingredients and a couple hours. DNA template Primers (anneal to flanking sequences) DNA polymerase dNTPs Mg2+ Buffer • Can be performed by hand or in a machine called a thermal cycler. • 1993: Nobel Prize for Chemistry
Fig. 9.3 Denature Anneal PCR Primers Extend PCR Primers w/Taq Repeat…
Bottom-up Mapping (Contig Maps, Mapping With Ordered Clones)
Whose genome did Celera sequence? Supposedly: African-American Asian-Chinese Hispanic-Mexican Caucasian Caucasian Actually: Celera’s genome is Craig Venter’s Science v. 291, pp 1304-1351
Cloning Vectors • Plasmid • Phagemid • Cosmid, fosmid • YACs • BACs • PACs
BAC Construction Scheme • BAC vector (pIndigo 536) T7 Insert SP6 Insert size ; 50-300Kb ( mean 110kb)
Application Of BAC library Whole genome sequencing Chromosome walking & contig assembly Fluorescent in situ hybridization BAC library Fine mapping for interest gene Construction of integrated genetic and physical map Positional cloning
“virtual autorad” - real-time DNA sequence output from ABI 377 Trace files (dye signals) are analyzed and bases called to create chromatograms. Chromatograms from opposite strands are reconciled with software to create double-stranded sequence data.
Maximum pyrosequencing read lengths currently are 300-500 nt. Commercial applications: 454 Life Sciences Genome Sequencer FLX Generate 400 million nt in 10 hours $5-7K USD per run $1M for mammalian genome
Real-time Quantitative PCR: Measures the abundance of DNA as it is amplified. Useful for quantitatively measuring the levels of mRNA in a sample. Uses reverse transcriptase to generate cDNA for the template. Can also be used to quantitatively estimate fraction of DNA from various organisms in a heterogenous sample (e.g, can be used to measure abundance of different microbes in soil sample). Fluorescent dye, SYBR Green, is incorporated into PCR reaction. SYBR Green fluoresces strongly when bound to DNA, but emits little fluorescence when not bound to DNA. SYBR Green fluorescence is proportional to the amount of DNA amplified, detected with a laser or other device. Experimental samples are compared to control sample with known concentration of cDNA.
Fig. 10.9 SYBR Green binds to double-stranded DNA and fluoresces