Understanding Valence Electrons and Types of Chemical Bonds in Physical Science
This comprehensive guide explores the concept of valence electrons and their significance in chemical bonding. It details the number of valence electrons in various elements belonging to Groups 1 to 8, discusses oxidation numbers, and explains the types of bonding: ionic, covalent, and metallic. Learn about how ionic bonds form between metals and non-metals, the nature of covalent bonds among non-metals, and the characteristics of metallic bonding in metals. Gain insight into practical examples for better understanding.

Understanding Valence Electrons and Types of Chemical Bonds in Physical Science
E N D
Presentation Transcript
Physical science notes
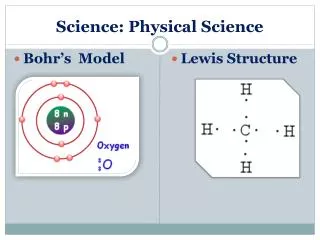
Valence electrons • Electrons on outer shell
Valence electrons • Group 1- 1 valence electron • Group 2- 2 valence electrons • Group 3- 3 valence electrons • Group 4- 4 valence electrons • Group 5- 5 valence electrons • Group 6- 6 valence electrons • Group 7- 7 valence electrons • Group 8- 8 valence electrons-except He (2)
Types of bonding • Ionic bond • Covalent bond • Metallic bond
Ionic bonds • An ionic bond is formed between a metal and a non-metal. Non-metals(-ve ion) are "stronger" than the metal(+ve ion) and can get electrons very easily from the metal. • These two opposite ions attract each other and form the ionic bond.
Common ionic bonds • NaCl • KBr • LiO
Covalent bonds • A covalent bond is formed between two non-metals that have similar electronegativities. Neither atom is "strong" enough to attract electrons from the other • For stabilization, they share their electrons from outer molecular orbit with others
Metallic bonding • Formed between atoms of metallic elements • Electron cloud around atoms • Good conductors at all states, lustrous, very high melting points • Examples; Na, Fe, Al, Au, Co
practice • What type of bond?
NaCl • CH4
answers • NaCl=ionic (metal+nonmetal) • CH4=covalent (2 nonmetals share e-) • Silver (Ag) below=metallic bond