Quantum Numbers Activity
Quantum Numbers Activity. Quantum Numbers. Quantum numbers tell us properties of the atomic orbitals, and electrons, in an atom. Like giving each electron its own addres

Quantum Numbers Activity
E N D
Presentation Transcript
Quantum Numbers • Quantum numbers tell us properties of the atomic orbitals, and electrons, in an atom. • Like giving each electron its own addres • An orbital is a 3-D region of an atom where there is a high probability of finding electrons (NOT an actual ring & NOT like the planetary orbitals)
Principal Quantum Number Angular Momentum Quantum Number Magnetic Quantum Number Spin Quantum Number
Textbook • You will need a textbook to look up information about the quantum numbers. Pg. 101.
Know for each Quantum # • Definition • Symbol • The allowed values • An example • A visual representation of the quantum number
Review Quantum Numbers as a Class Discussion of the 4 types of quantum numbers and there properties
Quantum Numbers • Used to describe various properties of the orbitals • Each electron is assigned a set of four quantum numbers which, in order, are n, l, ml, and ms • Like giving each electron its own address
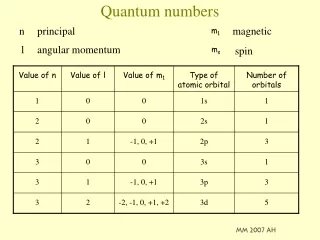
Principle quantum number • Definition: indicates the main energy level occupied by the electron • Symbol: n • Values: (written as integers) 1,2,3,4,5,6,7
Angular Momentum Quantum Numbers • Definition: indicates the shape of the orbital • Symbol: l • Values: • 0 = s • 1 = p • 2 = d • 3 = f • For a specific energy level, the number of orbital shapes available is equal to n -1
Magnetic Quantum Numbers • Definition: represents the orientation of an orbital around the nucleus • Symbol: ml • Values: for a p-orbital -1, 0 , 1
Spin Quantum Numbers • Definition: represents the spin states of electrons in an orbital • Symbol: ms • Values: +1/2, - ½
Can an e- be described by the following set of quantum numbers? n=2, l=1, ml= -1 All quantum numbers are allowed values
Can an e- be described by the following set of quantum numbers? n=1, l=1, ml= +1 Not possible. The value of l must be less than the value of n.
Can an e- be described by the following set of quantum numbers? n=7, l=3, ml= +3 All the quantum numbers are allowed values.
Can an e- be described by the following set of quantum numbers? n=3, l=1, ml=-3 Not possible. The value of ml must be in the range -l to +l
Replace the ? Mark with an appropriate quantum number. n=3, l=1, ml=? n=4, l=?, ml=-2 n=?, l=3, ml=?
Can an e- be described by the following set of quantum numbers? n=2, l=1, ml=-2 n=3, l=2, ml=+2 n=4, l=3, ml=+3 n=5, l=2, ml=+3
Writing Quantum Numbers • You can identify the quantum numbers for any electron in an element • Start by writing out the the electron configuration and orbital diagram for the element • Locate the electron in the orbital diagram that you are solving the quantum number for • Determine the 4 quantum numbers
Hydrogen’s electron • Write electron configuration • Draw orbital diagram • n= 1 • l= 0 • ml= 0 • ms= +1/2
Helium’s second electron • Write electron configuration • Draw orbital diagram • n= 1 • l= 0 • ml= 0 • ms= -1/2
Lithium’s third electron • Write electron configuration • Draw orbital diagram • n= 2 • l= 0 • ml= 0 • ms= +1/2
Flourine’s 6th electron • Write electron configuration • Draw orbital diagram • n= 2 • l= 1 • ml= 0 • ms= +1/2