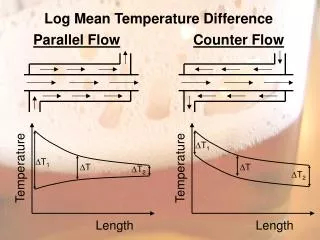
Log Mean Temperature Difference Parallel Flow Counter Flow
Log Mean Temperature Difference Parallel Flow Counter Flow. T 1. T 1. Temperature. Temperature. T. T. T 2. T 2. Length. Length. Log Mean Temperature Difference

Log Mean Temperature Difference Parallel Flow Counter Flow
E N D
Presentation Transcript
Log Mean Temperature Difference Parallel FlowCounter Flow T1 T1 Temperature Temperature T T T2 T2 Length Length
Log Mean Temperature Difference A tube-in-tube, counterflow heat exchanger carries hot wort at 85C in the inner tube and chilled water at 5C in the outer tube. The tube wall thickness is 4 mm and its thermal conductivity is 100 W/m.K. The wort film coefficient is 750 W/m2.K and the chilled water film coefficient is 3000 W/m2K. Determine the overall heat transfer coefficient and the rate of heat transfer per meter of heat exchanger length. Calculate the LMTD.
Fouling Layers of dirt, particles, biological growth, etc. effect resistance to heat transfer We cannot predict fouling factors well Allow for fouling factors when sizing heat transfer equipment Historical information from similar applications Little fouling in water side, more on product
Heat Exchanger Sizing • Beer, dispensed at a rate of 0.03 kg/s, is chilled in an ice bath from 18C to 8C. The beer flows through a stainless steel cooling coil with a 10 mm o.d., 9 mm i.d., and thermal conductivity of 100 W/m.K. The specific heat of the beer is 4.2 kJ/kg.K and the film heat transfer coefficients on the product and coolant sides are 5000 W/m2.K and 800 W/m2.K, respectively. The fouling factors on the product and coolant sides are 0.0008 and 0.00001 m2K/W. Assume that the heat exchanger is thin walled. • Determine the heat transfer rate • Determine the LMTD • Determine the overall heat transfer coefficient • Determine the outside area required • Determine the length of tube required
Heat Losses Total Heat Loss = Convection + Radiation Preventing heat loss, insulation Air – low thermal conductivity Air, good Water – relatively high thermal conductivity Water, bad Vessels/pipes above ambient temperature – open pore structure to allow water vapor out Vessels/pipes below ambient temperature - closed pore structure to avoid condensation
Heat Transfer – Continued Hot wort at 70C is transferred from one tank to another through a 2.0 cm id, 2.5 cm od stainless steel pipe (k = 120 W/m.K). The pipework is 21 m long and the wort has specific heat capacity of 4.0 kJ/kg.K and density of 975 kg/m3. The heat transfer coefficients on the inside and outside of the pipe are 4000 W/m2K and 12 W/m2K and the temperature of the surroundings is 18C. Assume that the pipe’s wall is “thin.” Calculate the rate of heat loss from the pipe and the exit temperature at the end of the pipe. How would adding a 1 cm layer of insulation (k = 0.05 W/m.K) to the outer surface of the pipe affect the exit temperature of the wort. Velocity in the pipe is 1.5 m/s.
Heat Transfer – Continued Previous Problem continued… Our pipe has an external emissivity of 0.7. Calculate the heat loss by radiation and compare it to the heat loss by convection.
Refrigeration Qout Condenser Compressor Win Evaporator Qin
Refrigeration 1-2: Constant entropy compression (s1 = s2) 2-3: Constant pressure heat rejection (3 = sat liq.) 3-4: Constant enthalpy throttling 4-1: Constant pressure heat addition (1 = sat vap.)
Refrigeration Efficiency = desired output / required input Desired output = Heat removal from refrigerated space (Qin) Required input = Work input to compressor Conservation of Energy: Qin + Win = Qout COP can be > 1.0 = Cooling Capacity
Refrigeration Applying Conservation of Energy…
Refrigeration Example • An ideal vapor-compression refrigeration cycle operates between the pressures of 10 and 1 bar. The system cools a secondary refrigerant at a rate of 25 kW. • Determine the mass flow rate of refrigerant. • Determine COP of the system. • Determine the power consumed by the compressor, in kW
Flash Pasteurization Minimum Safe Pasteurization Over Pasteurization 5.6 min Time (min) 0.1 1 10 100 Under Pasteurization 50 60 70 Temperature (C)
Flash Pasteurization Beer in = 0C Pasteurizer 60-70C 30 sec - 2 min 90-96% regeneration
Flash Pasteurization Pressure in Pasteurizer CO2 equilibrium pressure Pressure (Bar) Temperature (C) Temperature in Pasteurizer Time (sec)
Flash Pasteurization Typical Conditions: Beer inlet: 3C Outlet from regenerative heating: 66C Holding tube: 70C Outlet from regenerative cooling: 8C Outlet from cooling section: 3C Holding Time: 30 sec Advantages Little space required Relatively inexpensive equipment and operation Short time at “intermediate” temperatures where chemical changes occur without pasteurization
Tunnel Pasteurization Pasteurized after bottled or canned Bottles or cans move slowly down conveyer system Hot water sprays heat beer to pasteurization temperature Cool water sprays cool beer after pasteurization is complete Pressure builds in headspace - Volume of headspace - CO2 concentration in beer Bottles could break (Typical 1 in 500) CO2 could leak if bottles are not sealed well
Tunnel Pasteurization Spray water temperature Pressure (Bar) Temperature (C) Product Temperature Time (min)
Tunnel Pasteurization Simpler system than flash pasteurization Slow process (may take up to 40 minutes) Energy intensive process Beer near outside of can/bottle over pasteurized Mechanical failure, other stoppage could cause over pasteurization, effecting beer flavor