Understanding Refrigerators and Heat Engines: Efficiency and the Second Law of Thermodynamics
This lecture by Professor Lee Carkner explores the principles of refrigerators and heat engines, emphasizing their operational mechanics and efficiency. Key concepts include the relationship between pressure, volume, and temperature in thermodynamic cycles, the coefficient of performance of refrigerators, and the limitations imposed by the Second Law of Thermodynamics. The session highlights how work is utilized to move heat and offers insight into practical applications such as heat pumps and air conditioners, along with a thorough understanding of engine efficiencies and performance limitations.

Understanding Refrigerators and Heat Engines: Efficiency and the Second Law of Thermodynamics
E N D
Presentation Transcript
Refrigerators Physics 313 Professor Lee Carkner Lecture 13
Exercise #12 Engines • V1 = 6.25X10-4 m3, P1 = 12X106 Pa, n = 3 moles • P1V1g = P2V2g • P2 = P1V1g/V2g = 385 MPa • h = 1 – T1/T2 = 0.75 • W = hQH, QH = ncVDT23 • P3 = 500X106 Pa, V3 = 7.8X10-5 m3 • QH = (3)(3/2)(8.31)(1564-1205) = 13424 J
Limits on Engines • Engines convert heat into work and waste heat • Second Law of Thermodynamics • An engine cannot have 100% efficiency
1st and 2nd Laws • Converting heat completely into work does not violate the 1st law • The second law is an independent statement
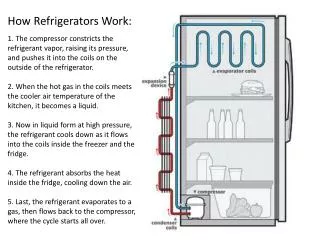
Refrigerators • A refrigerator is a device that uses work to move heat from low to high temperature • A heat pump does this to heat a room (want large QH)
How a Refrigerator Works • Fluid flows through the cold chamber and evaporates, adding heat QL to the fluid from the chamber • The fluid is pumped into the hot chamber and compressed, adding work W • The fluid condenses releasing heat QH
Refrigerator Cycle Compressor (work =W) Gas QL QH Low Pressure High Pressure Heat removed from fridge by evaporation Heat added to room by condensation Liquid Expansion Valve
Refrigerator Performance • The equivalent of efficiency for a refrigerator is the coefficient of performance K K = QL/(QH-QL) • Unlike efficiency, K can be greater than 1
Air Conditioner • Air conditioners also have condensers that dry out the air
Heat Pump • The heat removed from the inside of a refrigerator is ejected into the kitchen • A refrigerator that has the cold chamber as the outdoors and the hot chamber as the house is called a heat pump • Many heat pumps can be reversed in summer to function as air conditioners
Refrigerators and the Second Law • You cannot move heat from low to high temperature without the addition of work
Statements of the Second Law • Kelvin-Planck Statement: • Clausius Statement:
Equivalence • One implies the other • For example: • A 100% efficient engine connected to a high T reservoir powering a refrigerator cooling a low T reservoir to the same high T reservoir • The refrigerator by itself is “legal” but the net effect to is move QL from low to high T with no other effect
Engines and Refrigerators • Efficiency: h = W/QH = (QH-QL)/QH = 1 - (QL/QH) • Can rewrite using: • Coefficient of performance: K = QL/W = QL/(QH-QL) (refrigerator)