Predicting Toxicity in Advanced Gastrointestinal Stromal Tumor Patients Treated with Imatinib
This study evaluates the toxicity of imatinib mesylate in patients with advanced Gastrointestinal Stromal Tumors (GIST) through a randomized phase III trial involving 946 patients. It compares the effects of daily doses of imatinib (400 mg once daily vs. 400 mg twice daily) over a median follow-up of 42 months. Using univariate and multivariate regression models, the study identifies prognostic factors for grade 3-4 anemia, neutropenia, and other toxicities. An interactive risk calculator is provided to aid clinicians in predicting toxicity based on individual patient data.

Predicting Toxicity in Advanced Gastrointestinal Stromal Tumor Patients Treated with Imatinib
E N D
Presentation Transcript
EORTCSTBSG Predicting toxicity for patients with advanced Gastrointestinal Stromal Tumors (GIST) treated with imatinib mesylate : an EORTC/ISG/AGITG randomized trial M. Van Glabbeke, J. Verweij, P.G. Casali, J. Simes,A. Le Cesne, P. Reichardt, R. Issels, J.R. Judson,A.T. van Oosterom, J.-Y. Blay
Material • Randomized phase III study comparing daily doses of imatinib (400 mg od vs 400 mg bid) for the treatment of patients with advanced GIST • 946 patients randomized • 942 patients started therapy • 310 patients still under therapy • Follow-up • Median: 42 months (3.5 years) • At 1, 2, 3 and 4 years: 99%, 90%, 81% and 4%
Methods • Prognostic factors analyses • End-points (binary) : occurrence of • Grade 3-4 anemia, neutropenia • Grade 2-4 edema, fatigue, nausea, diarrhea, skin rash • Investigated co-factors • Imatinib dose • Age, sex, PS, baseline laboratory parameters • Time since GIST diagnosis, site of origin, lesion size • Prior therapies • Logistic univariate / multivariate regression models • Univariate model: select variables if P < 0.05 • Multivariate model: keep variable if P < 0.01
Cumulative incidence of toxicities(non hemato.grade 2+, hemato. grade 3+) 400 mg o.d. 400 mg b.i.d. Edema - Skin rash - Fatigue - Nausea - Diarrhea Anemia - Neutropenia
Multivariate models : non hematological grade 2+ toxicities ++ : P < 0.01, --- : P < 0.001, +++ : P < 0.001, ++++ : P < 0.0001
Multivariate models : hematological grade 3+ toxicities -- : P < 0.01, ---- : P < 0.0001 , ++++ : P < 0.0001
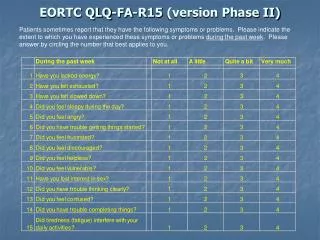
Interactive risk calculator www.eortc.be / tools / imatinibtoxicity • Interactive version • Downloads(Excel) • For PC • For PDA
Interactive risk calculator for predictingimatinib toxicities in GIST patients
Validation • Validation data set • EORTC phase I-II trial of imatinib • 91 patients with advanced or metastatic sarcoma • Daily doses: 400 mg, 600 mg, 800 mg, 1000 mg • Patients classified in “risk group” according to probability of each toxicity estimated with the model • < 20%, 20-40%, > 40% or < 20%, > 20% • Comparison with the observed proportion of events • Within the expected range for 10/13 risk groups (> 10 pts) • Overlap with the 95% confidence interval in all groups • Underestimation of edema & nausea (low risk groups) • Underestimation in male patients ?
Conclusion (I) • All toxicities are highly dose dependent, except neutropenia • Anemia and neutropenia only depend on baseline hematological factors • Risk of non hematological toxicity increase with age, poor PS and in female • Confirmation of other series • Other factors • Prior chemotherapy (fatigue) • Small lesions (rash) • Identified GI origin (diarrhea)
Conclusions (II) • Interactive risk calculator to estimate the risk of toxicity for individual patients • May be used in the clinic • Available on the internet (Excel spreadsheet) • May be used to customized treatment ? • Validation • Based on a limited data set • Underestimate of edema and nausea in male patients ? • Should be validated on larger data series