Comprehensive Review of Endocrine Principles and Hormonal Functions
E N D
Presentation Transcript
6. ENDOCRINE PRINCIPLES V BS 122 2012 Luis A. Bate
OBJECTIVES TO REVIEW BASIC ASPECTS OF ENDOCRINOLOGY TO ESTABLISH A BASELINE KNOWLEDGE REFERENCE Review Chapter 5 Senger Chapter 74 Guyton
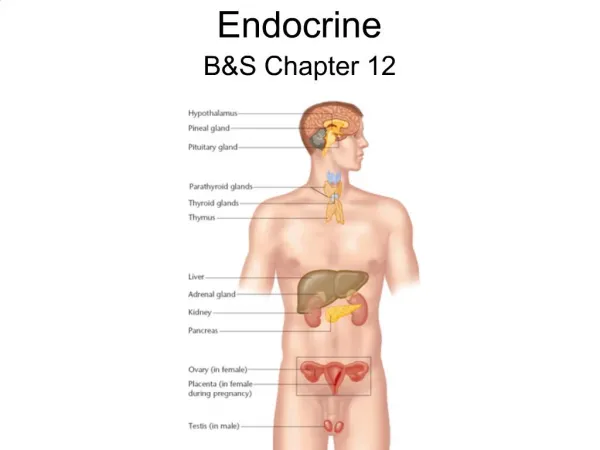
HORMONES Potent regulators of biological functions Their biosynthesis, storage, excretion and metabolism depends on their chemical structure and composition F 6-1
NEURO-ENDOCRINE NERVOUS ENDOCRINE NEURON SECRETORY CELL NEURON TARGET TARGET TARGET F 6-2
AUTOCRINE SECRETORY CELL ENDOCRINE PARACRINE TARGET TARGET F 6-3
CLASSIFICATION • Origin • Pituitary, thyroid, gonad • Activation • Direct synthesis, conversion, cleavage • Chemical composition • Amino acids, lipids, proteins • Function/action • Neurotransmitters, inhibitors F 6-4
TYPE OF HORMONES Amino acid derivatives Proteins Lipid derivatives F 6-5
AMINO ACIDS DERIVATIVES • Neurotransmitters • Catecholamines (Tyrosine derivative) • Epinephrine • Norepinephrine • Dopamine • GABA • Thyroid hormones • Triiodothyronine (T3) • Thyroxine (T4) F 6-6
PROTEINS • Small peptides • TRH 3 aa • Polypeptides • GnRH 10 aa, ACTH 41 aa • Glycoproteins • LH, FSH, TSH F 6-7
LIPIDS • Steroids • Progestogens, glucocorticoids • 21 C progesterone, cortisol • Androgens • 19 C testosterone • Estrogens • 18 C estrone, estradiol, estriol • Prostaglandins • PGF2α, PGE, PGH F 6-8
Glutamate HOOC CH (CH ) COOH (CH ) COOH 2 2 2 3 decarboxylase NH NH 2 2 HYPOTHALAMUS CO GLUTAMICACID GABA 2 ( GAMMA-AMINO BUTYRIC ACID) F 6-9
O NH NH 2 2 CH CH CH CH HO 2 2 COOH COOH O TYROSINE PHENYLALANINE CO 2 2 NH 2 NH NH 2 2 CH CH HO 2 CH CH CH CH HO HO 2 2 2 2 COOH HO DOPA DOPAMINE HO DOPAMINE O B-OXIDASE H CH 2 CH H N 3 H NH 2 C CH HO HO C CH 2 2 PNMT O O (Phenyl ethanol H H amine N HO HO Methyl transferase) EPINEPHRINE NOREPINEPHRINE ADRENAL GLAND RATE LIMITINGSTEP F 6-10
Cys OXYTOCIN Tyr S IIe S Gln Cys-Pro-Leu-GlyNH 2 Asn Cys Tyr S IIe S Gln Cys Pro-Leu-Gly NH Asn 2 MIF-I TOCINOIC ACID MIF-II F 6-11
INSULIN INSULIN PROINSULIN CPEPTIDE αCHAIN βCHAIN F 6-12
β α Active hormone β unit Inactive α unit inactive F 6-13
β unit LH properties α unit common β unit FSH properties F 6-14
NH 2 CH CH COOH O HO 2 T 4 NH 2 CH CH COOH HO O 2 ACTIVE T 3 NH 2 CH CH COOH O HO 2 INACTIVE rT 3 NH 2 O CH CH COOH HO 2 THYRONINE T CATABOLITE 0 THYROID RELATEDHORMONES PROHORMONE? F 6-15
Cyclopentanopherhydrophenanthrene ph ta no er hy en Cy cl op dr op • e he nt en na hr F 6-16
CHOLESTANE 21 CH CH 27 3 3 18 20 CH CH CH CH 12 CH H C 2 2 2 22 23 24 25 3 17 11 16 CH 13 D 26 3 C 19 1 H C 3 9 14 15 2 10 8 A B 3 7 5 4 6 F 6-17
CH CH 3 3 CH CH CH CH CH H C 2 2 2 3 CH 3 H C 3 F 6-17
CH 3 CH CH CH CH CH 3 2 2 2 CH CH H C 2 3 3 HC 3 CHOLESTANE TO PREGNANE = 21 C F 6-18
CH 3 CH 2 H C 3 HC 3 PREGNANE TO ANDROSTANE = 19 C F 6-19
H C 3 H C 3 ANDROSTANE TO ESTRANE = 18 C F 6-20
STEROID NOMENCLATURE • Prefix Suffix Indicates • Hydroxy -ol Hydroxyl group (-OH) • β-OH - Hydroxyl above plane • α-OH - Hydroxyl below plane • Oxo one Keto or carbonyl group (C=O) • - al Aldehyde (-CHO) • Carboxy -oic acid Carboxylic acid (COOH) • - -ene Double bond (-C=C-) • - -yne Triple bond (-C=C-) • - -ane Saturated ring
CORTISOL 21 OH CH 2 20 C O HC OH OH 3 17 11 H C 3 3 O 4 PREGN-4-ENE-3, 20 DIONE, 11β, 17α, 21 TRIOL F 6-22
PROSTAGLANDINS Arachidonicacid Cyclooxygenase Prostaglandin E F 6-23
PROSTAGLANDINS F 6-24
LATENT PERIOD • Ultra short Pre-existing , Enzymes (Seconds) Permeability • Very short Enzymatic cascades (Minutes) • Short Peptide assembly (½-1 hour) • Long Protein induction (Hours) • Very long Growth, cell proliferation (Days) • Ultra long Cell enhancement (Weeks) F 6-25
β α γ G protein F 6-26
SECOND MESSENGER IAC AAC α β γ G protein DIRE CAMP ATP IPK APK Response: Enzyme act Membrane change Prot Synth. PARE ATP ADP F 6-27
CLASSICAL STEROID ACTION F 6-28
RECEPTOR Structure to which a hormone binds to trigger a biological response It is composed of a binding site joined by a transduction or coupling mechanism to an effector site F 6-29
Characteristics of G Protein- Coupled receptors Extra cellular amino terminal variable number of aa 3 extra cellular loops (exoloops) 7 transmembrane domains 20-27aa 3 intracellular loops (introloops) Intracellular carboxyl terminal 10-700 aa F 6-30
G protein Channel Enzyme Nuclear receptor RECEPTOR TYPES F 6-31
HORMONE SPECIFICITY Non Specific= No Response Specific=Response Semi Specific= Some Response Specific= Response F 6-32
RECEPTOR AFFINITY Highaffinity Lowaffinity Response Response F 6-33
NUMBER OF SITES Response No response F 6-34
SPECIFICITY OFTISSUE No response Response F 6-35
HORMONE Synthesis Secretion Transport Metabolism RECEPTOR Synthesis Modification Metabolism Regulation of hormone availability and biological activity[H] + [R] ↔ [HR] F 6-36
DOWN-REGULATION Full response Low response F 6-37
UPREGULATION Modest response Maximal response F 6-38
HETEROREGULATION F 6-39
NEGATIVE FEEDBACK F 6-40
POSITIVE FEEDBACK F 6-41