Understanding Acid-Base Titrations: Calculations and Techniques
This lesson covers the principles of acid-base titrations, focusing on how to determine molarity through the reaction between a known concentration acid and a base. It discusses essential lab equipment, including burettes and volumetric flasks, and provides step-by-step calculations for titration experiments. Two examples illustrate how to calculate unknown base concentrations based on titration results and how to prepare a standard solution of H2C2O4. This lesson also emphasizes proper data handling and averaging results from multiple trials for accurate outcomes.

Understanding Acid-Base Titrations: Calculations and Techniques
E N D
Presentation Transcript
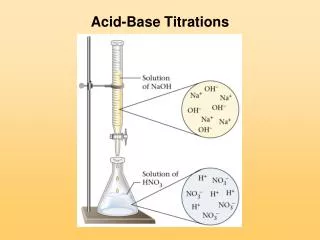
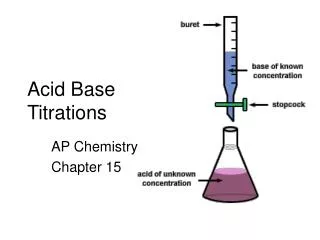
Acid Base Titrations Lesson 4
In a titration the molarity of one chemical is determined by reacting it with another one with known molarity (standard).
Special Equipment is used for a titration Burette Erlenmeyer Flask Magnetic Stirrer Pipette Pipette Filler
Digital Balance Solid acid to make a standard solution Volumetric Flask
Titration Calculation 1. 6.50 mL of 0.100 M H2C2O4 is required to neutralize 10.0 mL of KOH solution in a titration. Calculate the base concentration. H2C2O4 + 2KOH K2C2O4 + 2H2O 0.00650 L 0.0100 L 0.100 M ? M 0.00650 L H2C2O4 x 0.100 mole x 2 mole KOH 1 L 1 mole H2C2O4 [KOH] = 0.0100 L = 0.130 M
If you get data in atable, you need to subtractthe finalburette reading from the initialto get the volumeof acid or base added. If you have multiple trials, averagethem but reject any volumes that are way off. Use some common sense. Burette Volume in mL Initial 0.00 mL 8.95 mL 17.41 mL Final 8.95 mL 17.41mL 25.85 mL Volume Added8.95 mL 8.46 mL 8.44 mL reject average 8.46 + 8.44 = 8.45 mL 2
Titration Calculation 2. 8.95 mL , 8.46 mL and 8.44 mL of 0.200 M H2SO4 was required to neutralize 25.0 mL of KOH solution in a titration. Calculate the base concentration. H2SO4 + 2KOH K2SO4 + 2H2O 0.00845 L 0.0250 L 0.200 M ? M [KOH] = 0.00845 L H2SO4 x 0.200 mole x 2 mole KOH1 L 1 mole H2SO4 0.0250 L = 0.135 M
3. Calculate the mass of H2C2O4.2H2O required to make 100.0 mL of a 0.1000 M standard solution to use in your titration lab tomorrow. 0.100 L x 0.1000 mol x 126.06 g = 1.261 g 1 L 1 mole