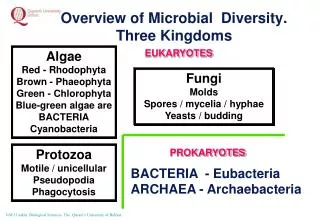
Microbial Diversity
Microbial Diversity. Morphological. Not much to see!. Morphological Diversity. Microbial Diversity – Genetic. Microbiology!!!. Functional Diversity. Biochemical/metabolic E.g. Rhodoferax ferroreducens Microbial fuel cells Pass electrons to anode, produce power from waste

Microbial Diversity
E N D
Presentation Transcript
Microbial Diversity • Morphological Not much to see!
Microbial Diversity – Genetic Microbiology!!!
Functional Diversity • Biochemical/metabolic • E.g. Rhodoferax ferroreducens • Microbial fuel cells • Pass electrons to anode, produce power from waste • E.g. Vibrio cholerae • Severe diarrhea Rhodoferax ferroreducens Vibrio cholerae
Habitat Diversity • Environmental conditions • Where in the world? • Other worlds? Yellowstone
Microbial Diversity – Overview • Oxygen requirements (metabolism) and tolerance (habitat) • Nutritional diversity (metabolic) • Carbon, energy, and electron sources • Habitat diversity • Extremophiles
Oxygen • Toxic for some • Requirement for some • Like us!
Metabolism Catabolism Anabolism Provides energy for Uses Uses Degradative pathways Biosynthetic pathways Both bi-directional Amphibolic pathways
Catabolism Overview • Humans, animals, many microbes
A bunch of steps!!! Catabolism:Oxidation-Reduction C6H12O6 • Electron source (reduced) • Aka “energy source” • Electron acceptor (oxidized) • Aka “terminal electron acceptor” (TEA) • End products: • Oxidized electron source • Reduced electron acceptor + O2 CO2 + H2O
Glycolysis • The oxidation of glucose to pyruvic acid, produces 2 ATP and 2 NADH+ 2 ATP + 2 NADH+
PreparatoryStage Glucose Glucose 1 • 2 ATPs used Glucose 6-phosphate Fructose 6-phosphate Fructose 1,6-diphosphate Glyceraldehyde 3-phosphate (GP) Dihydroxyacetone phosphate (DHAP) Figure 5.12.1
6 1,3-diphosphoglyceric acid 7 3-phosphoglyceric acid 8 2-phosphoglyceric acid 9 Phosphoenolpyruvic acid (PEP) 10 Pyruvic acid Figure 5.12.2 EnergyProducing Stage • 4 ATP produced • 2 NADH+ produced
Overall: Glucose + 2 ADP + 2 PO4– + 2 NAD+2 pyruvic acid + 4 ATP + 2 NADH + 2H+
Intermediate Step • Pyruvic acid is oxidized and decarboyxlated Figure 5.13.1
Krebs Cycle • Aka: • TCA cycle • Citric acid cycle • 1 ATP • 2 NADH+ • 1 FADH2 Figure 5.13.2
Respiration • Oxidation liberates electrons – used to produce NADH+ and FADH2 • Electrons then passed along an electron transport chain (ETC) • ATP generated by oxidative phosphorylation • Proton motive force • ATPase • 1 glucose 38 ATP
Electron Transport Chain • Proton motive force • Proton gradient • Ion gradient • PMF = DpH + DY
Respiration • Aerobic respiration: TEA is O2 • Anaerobic respiration: TEA is not O2 • Yields less energy than aerobic • Why?
Anaerobic Rs E0’ = electropotential (Voltage) DG aDE0’ The greater the difference in E0’ between e- source and TEA, the more energy available
Aerobic Vs. Anaerobic • E. coli • Which produces more PMF? • Why?
Chemoorganotrophs Diversity: Bacteria, Archaea vs. Eukarya
Fermentation • Produces ATP only from substrate-level phosphorylation (SLP) • Far less energy than Rs • 1 glucose 2 ATP • Does not use Krebs cycle or ETC for energy generation • Uses an organic molecule as the terminal electron acceptor • Need to recycle the NAD+
Types of Fermentation • Eukaryotes and prokaryotes
Types of Fermentation Figure 5.18b
Laboratory Tests Labs 3C and 8
Metabolism Switchers • Human cells, yeast • Aerobic Rs and Fm • Pseudomonas aeruginosa • Aerobic Rs and Rs of NO3- • E. coli • Aerobic Rs, Anaerobic Rs, Fm
Oxygen Requirement Diversity Knows these terms!
Oxygen Thioglycolate broth resazurin
Culturing Anaerobes Anaerobic glove box Gas Pack
Toxicity of Oxygen • We live in a toxic world! • Atmosphere full of a toxic, explosive gas: O2 • Every breath generates chemicals that damage membranes, oxidize macromolecules (such as DNA) and kill cells • Aerobes, such as ourselves, must detoxify!
Toxic Forms of Oxygen • O2 – already very reactive • Singlet oxygen (electrons shifted to higher levels) • Created photo- and bio-chemically • Carotenoids protect cells
Toxic Forms of Oxygen • Superoxide anion (O2-) and hydrogen peroxide (H2O2) • Produced as by-products during respiration • Hydroxyl radical (OH.) • Produced by ionizing radiation (e.g. UV) • Extremely reactive • Antioxidant molecules can protect • E.g. carotenoids
Enzymatic Protection Lab 3 Staphylococcus Streptococcus
Obligate Aerobes • Usually • Catalase+ • Peroxidase+ • SOD+ • E.g. • Most eukaryotes • Azotobacter • Many Bacillus sp.
Microaerophiles • Usually • Catalase- • Peroxidase+ • SOD - variable • E.g. • Helicobacter pylori • Magnetospirillum magnetotacticum
Facultative Aerobes • Usually • Catalase+ • Peroxidase+ • SOD+ • E.g. • E. coli • Shigella • Pseudomonas aeruginosa
Aerotolerant Anaerobes • Strictly fermentive metabolism • Can grow in presence of azide • Usually • Catalase- • Peroxidase+ • SOD+ • E.g. • LAB • Streptococcus, Enterococcus, Lactobacillus
Obligate Anaerobes • Usually • Catalase- • Peroxidase- • SOD- • E.g. • Clostridium (fermenter) • Desulfovibrio vulgaris (anaerobic Rs)
Lab Tests • Catalase • Oxidase (tests for cytochrome c) • EMB and MacConkey agar (lactose fermentation, acid production) • Lactose and glucose broth • Azide broth, KF agar