Revolutionizing Medical Reporting: DICOM SR & CDA Rel.2
80 likes | 123 Vues
Explore the integration of DICOM SR and CDA Release 2 for structured and coded medical reporting, enhancing decision support, EHR integration, and data analysis. Discover the strategic insights shaping the future of medical reporting. Proposed actions and initiatives highlight advancements in the field.

Revolutionizing Medical Reporting: DICOM SR & CDA Rel.2
E N D
Presentation Transcript
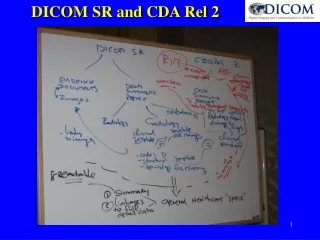
DICOM SR and CDA Rel 2 • Summary Imaging Report (SIR) • Patient Personal Record • Back to Referring Physician SIR SIR is extract of Imaging Report Specialized Diagnostics & Intervention • Mammo • US • Cardiac • Ophthalmology Imaging Reports Images Evidence Docs • Specialist • Evidence = Structured Info • Images = Structured Info Object referenceUse/create info content
We had a closer look at one of our main topics: Structured reporting objects and templates, relationship with CDA Release 2 Actions: Update the Policy Paper on Structured Documents, also including IHE in discussion.Kevin, Niki, Charles, Cor. Review by Lloyd, John, Joel. Have telcon with WG20/WG10/WG6 about Radiology report workitem: scope, purpose, DICOM’s help in adoption process, collaboration with HL7.Also: DICOM encapsulated CDA, sup 114 Strategic process
1. Today in the clinical practice we hardly see any application of structured and coded reporting beyond the evidence documents. The expectation and vision is that the need for structured and coded reporting will emerge the coming years to facilitate decision support, full integration in the patient’s EHR, data mining and analysis to improve the theoretical foundation and practice of medicine. 2. The workload for creation of structured and coded reports is the most significant acceptance criteria for the reporting clinician. There should be a direct link between the guidelines and practice protocol and the template/document structure. 3. The expectation is that given the broad range of clinical domains, broad scope of the use cases that need to be supported, and IT infrastructures and applications that are in place today, there will be a coexistence of both DICOM SR and HL7 CDA for encoding structured semantics. Strategic Insights
4. The reporting use cases identify the need to exactly capture the report layout. This is legally required in certain jurisdictions, and because of human perception and readability, and business presentation and reimbursement. PDF is well suited to support these requirements. 5. The assumption is that in some/many reporting use cases the reference to the evidence documents and images from the report is an important element in the report. 6. The assumption is that the report and the referenced evidence documents and images have to be incorporated in the patient's electronic health record. 7. Reaching agreement on clinical templates for specific applications is a significant challenge to the adoption of structured and coded reporting, both for the professional societies to generate and the IT infrastructure in the practice to be flexible enough to deal with these. Strategic Insights
a. In the DICOM community we need to find and document at least one typical clinical use case where we believe DICOM SR is ideally suited to encode the structured report, and efficiently supports the whole scope of the use case including the verification, distribution and medical research. The DICOM standard has all the elements in place to support such an "all DICOM" structured reporting use case. [1. Breast Imaging Report, 2. Echo] b. DICOM should communicate to the IHE community it’s acceptance of the coexistence of DICOM and HL7 constructs for the purpose of structured and coded reports. Proposed Actions
c. WG20 is on the right track with the ongoing effort to harmonize the structured report content between DICOM SR and HL7 CDA, because in practice it may well be that it is more the hospital's IT and application infrastructure that is the decisive factor for the choice of structured report encoding, rather than the clinical use case. WG20 will be supported in their activity to define a Diagnostic Report template by the activity defined in action a. d. Because DICOM has a method for encapsulating PDF, IHE is encouraged to incorporate into the reporting workflow a mechanism by which DICOM SR is complemented by an equivalent encoding of the report in PDF. e. Advise to the DICOM Committee to set-up a liaison with Physician’s EHR Coalition for the topic of clinical guidelines and the relationship to structured and coded reporting. Proposed Actions