Experiment 20:
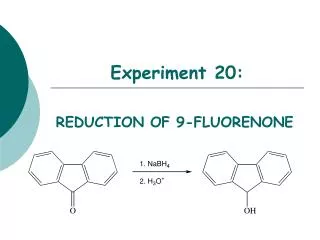
Experiment 20:. REDUCTION OF 9-FLUORENONE. Objectives:. To synthesize a secondary alcohol from a ketone using a sodium borohydride reduction. To purify and isolate the product using recrystallization. To analyze the purity of the product using TLC, HPLC and melting point analysis.


Experiment 20:
E N D
Presentation Transcript
Experiment 20: REDUCTION OF 9-FLUORENONE
Objectives: • To synthesize a secondary alcohol from a ketone using a sodium borohydride reduction. • To purify and isolate the product using recrystallization. • To analyzethe purity of the product using TLC, HPLC and melting point analysis. • To characterize the reactants and products using NMR spectroscopy.
Before coming to lab… • Review these techniques: • Fractional distillation • Vacuum filtration • GC Analysis
REDUCTION USING NaBH4 • NaBH4 (sodium borohydride) is a versatile and useful reducing agent in organic chemistry. • A reducing agent causes a reaction (a reduction) in which the product has more bonds from carbon to hydrogen (or fewer bonds to oxygen)
MECHANISM NaBH4 transfers a hydride ion to the carbonyl carbon. The oxygen anion eventually removes a proton from water.
EXPERIMENTAL PROCEDURE(Synthesis) • Weigh 9-fluorenone into 50 mL flask with stir bar. • Dissolve in ethanol. • Add NaBH4 slowly. Stir 20 minutes. • Add deionized water. • Add 0.1 M HCl dropwise until pH = 7.
EXPERIMENTAL PROCEDURE(Purification) • Suction filter. • RINSE SOLID WITH 10 ML COLD WATER! • Prepare TLC and HPLC samples. • Place product in warm oven for 10 minutes on a preweighed watch glass. • Obtain final product yield, calculate % yield. • Proceed to PRODUCT ANALYSIS.
EXPERIMENTAL PROCEDURE(Product Analysis) • TLC analysis • Identify components present in product solution and determine purity. • HPLC analysis • Identify components present in product solution and determine purity. • MP analysis • Compare to literature value of melting point to determine purity. • NMR analysis • Characterize reactant and product using spectra on page 169.
NMR Spectroscopy9-fluorenone Aromatic protons Aromatic carbons NMR solvent 194d
NMR Spectroscopy9-fluorenol Aromatic protons 5.54d TMS 1.75d Aromatic carbons NMR solvent 75d
SAFETY CONCERNS • All compounds used in today’s experiment are FLAMMABLE • and TOXIC! Use extreme caution when in use!
WASTE MANAGEMENT • Place aqueous filtrate from suction filtration in container labeled “AQUEOUS WASTE (Ketones)”. • Place TLC solvent in container labeled “ORGANIC WASTE (Ketones)”. • Place MgSO4, filter papers, and used TLC plates in yellow “SOLID WASTE” can. • Place used TLC and melting point capillaries in BROKEN GLASS CONTAINER. • Place product in container labeled “9-FLUORENOL (Student Prep).
CLEANING • Clean all glassware with soap, water, and a brush if necessary. • Rinse all glassware with wash acetone before returning it to the lab drawer! • DO NOT return glassware to lab drawer dirty or wet!