Exploring BioA as a Target for Antibacterial Drug Development Against Mycobacterium tuberculosis
Target-based antibacterial drug discovery faces numerous challenges, particularly in identifying viable targets. This study investigates the potential of BioA, a critical enzyme in the biotin biosynthesis pathway of Mycobacterium tuberculosis (Mtb), as a drug target. Using conditional knockdown mutants, we explore the impact of silencing bioA during Mtb infections. Insights into the vulnerability of Mtb to partial inactivation of BioA will guide the development of small molecules that can inhibit this enzyme effectively, with implications for acute and chronic infections.


Exploring BioA as a Target for Antibacterial Drug Development Against Mycobacterium tuberculosis
E N D
Presentation Transcript
Modeling bacterial phenotypes using conditional knockdown mutants Dirk Schnappinger, PhD Weill Cornell Medical College
Target-based antibacterial drug discovery has been difficult Payne et al (2007) Drugs for bad bugs: confronting challenges of antibacterial discovery. Nature Reviews Drug Discovery 6, 29.
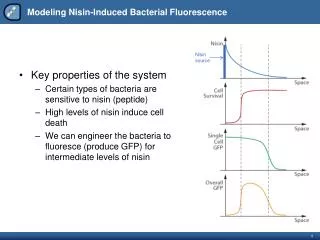
Target-based antibacterial drug discovery – some reasons for failure • Selection of an inappropriate target → Develop and apply a genetic approach to identify Mtb genes required for growth and survival during all phases of an infection. → Measure vulnerability of Mtb to partial inactivation specific enzymes. • Biochemical screens against purified enzymes do not select for compounds that are able to enter the bacterial cell → Engineer Mtb strains that are hypersusceptible to inhibition of a specific enzyme or pathway.
Evaluating Mtb proteins / pathways as new targets for drug development: biotin metabolism • Biotin is required to synthesize several essential components of the mycobacterial cell envelope. • Transposon mutants are strongly attenuated in mice (Sassetti and Rubin).
The biotin biosynthesis pathway Amiclenomycin prevents mycobacterial growth through inhibition of BioA. Crystal structure of BioA has been solved (Sacchettini).
MtbDbioA Biotin concentration in human serum: 0.1 to 3.3 nM (Hansen & Holm, Clin. Chem. 35/8, 1989) 0.01 to 0.2 nM (Hayakawa & Oizumi J Chrom 1987)
Impact of silencing bioA during infections Plus doxy Doxy day 1 to 10 Doxy day 1 to 56 Day 56 Dav 112 Day 168 Day 224
Conclusions relevant to the evaluation of BioA (biotin metabolism) as a drug target Small molecules that efficiently and specifically inhibit BioA are predicted: • to be inactive in the presence of >25 nM biotin, • to be bactericidal in the absence of biotin, • to be effective during acute and chronic infections (given sufficient bioavailability), • to require months to eliminate Mtb during an infection. Correlation between chemistry and genetics will of course not be perfect.
How efficient do BioA inhibitors have to be? MtbbioA TetON-1 grows without inducer in mice.
Engineer Mtb strains that are hypersusceptible to inhibition of a specific enzyme or pathway
Engineer Mtb strains that are hypersusceptible to inhibition of a specific enzyme or pathway bioATetON-1 M. tuberculosis
Summary BioA is required for (i) growth and survival of Mtb in biotin-free liquid medium, and (ii) growth and persistence of Mtb in mice. BioA is a low vulnerability target but an otherwise “druggable” target and partial knockdown mutants might facilitate the development of BioA inhibitors with whole cell activity. Developed system with improved gene silencing activity and faster kinetics of inactivation.
Weill Cornell Ehrt, Sabine Ferraras, Julian Klotzsche, Marcus Monteleone, Mercedes Odaira, Toshiko Park, SaeWoong Thanks to University of Minnesota Aldrich,Courtney Finzel, Barry Duckworth, Benjamin Shi, Ce Wilson, Daniel Novartis, Singapore Camacho, Luis Dartois, Veronique Dick, Thomas Manjunatha, Ujjini Pethe, Kevin Rao, Srini NIH/NIAID Barry, Clifton Boshoff, Helena University of Pittsburgh Flynn, JoAnne Lin, Philana Imperial College Robertson, Brian Williams, Kerstin Robert Wilkinson Young, Douglas Harvard University Rubin, Eric Wei, Jun-Rong Stanford Dolganov, Gregory Schoolnik, Gary Yonsei/Masan, Korea Cho, Ray Taek-Sun Song SBRI Rustad, Tige Sherman, David