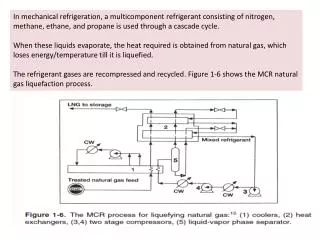
Gas Properties
440 likes | 1.13k Vues
Gas Properties. Gasses. A state of matter characterized by: Filling the container it occupies Having a volume that is not fixed Particles that have little or no intermolecular forces acting on them Particles have freedom of motion. Pressure. Pressure – Force per Area

Gas Properties
E N D
Presentation Transcript
Gasses • A state of matter characterized by: • Filling the container it occupies • Having a volume that is not fixed • Particles that have little or no intermolecular forces acting on them • Particles have freedom of motion
Pressure • Pressure – Force per Area • Atmospheric pressure is 14.7 lbs/in2 • Pressure is caused by the force of molecules striking a surface
Pressure • Typical Units Include • Atmospheres (atm) • Kilopascals (kPa) • Millimeters of mercury (mmHg) • Torr (torr) • 1atm = 101.325 kPa = 760 mmHg = 760 torr
Temperature • Relative measure of • Translational energy • Movement in a particular direction • Vibrational Energy • Rotational Energy • If two substances are at the same temperature, no NET energy is transferred between them.
Temperature Conversions • Convert 25°C to Kelvin • Convert 345K to Celsius • Convert -45°C to Kelvin • Convert 288K to Celsius
Properties of Gases • The properties of gases are interdependent. • Pressure (P) • Volume (V) • Temperature (T) • Amount (moles, n) • Changing one can affect the others • Exception - The amount of gas affects the other variables but the other variables do not affect the amount
Animated Gas Lab • http://www.grc.nasa.gov/WWW/k-12/airplane/Animation/frglab2.html • Hold two variables constant • Volume • Amount of gas • What is the relationship between pressure and temperature? • As temperature increases, pressure increases • Direct Relationship
With Numbers Pressure (atm) • A stoppered flask in a hot water bath is allowed to cool along with the bath. Temperature (°C)
Pressure vs. Temperature • Calculate the x-intercept • When P = 0… • That’s very close to -273 °C
Pressure vs. Temperature • To avoid having the x-intercept not at the origin… • We define absolute temperature – Kelvin • K = °C + 273 • This defines an absolute zero • Absolute zero - (0K) - the temperature where a substance is in its lowest possible energy state. • Always use Kelvin in Gas Law Problems
Pressure vs. Temperature P = (constant) x T
Racquet ball • Liquid_Nitrogen_-_Racquet_Ball.avi • Think of the ball as a container of gas – balls bounce because they have a pocket of gas inside • Use liquid nitrogen to cool a racquet ball • What changes?
Plunger Fun • Monitor the pressure in a container while the plunger is moved in and out. • http://www.grc.nasa.gov/WWW/k-12/airplane/Animation/frglab2.html • What is held constant? • Pressure? • Volume? • Temperature? • Amount of gas? • What is the relationship between pressure and volume? • As the volume increases, pressure decreases • Inverse Relationship
Injecting Air Into a Sealed Container • What is held constant? • Pressure? • Volume? • Temperature? • Amount of gas? • What is the relationship between pressure and amount of gas? • As the amount of gas increases, pressure increases • Direct Relationship
Pressure vs. Moles (n) • P = (constant) x n
Can Activity • Put water in the soda can to cover the bottom surface. • Heat the can on the hot plate until you can see steam. • With the tongs, QUICKLY turn the can upside down and immerse it in the cold-water bath. • Record your observations.
Can Activity • Explain your results in terms of P, V, n, and T. • Where did all the water in the can come from? • What do you think would happen if you did this experiment with a glass jar instead of a can? • What do you think would happen if you did this experiment with an empty can? • What do you think would happen if you did this experiment with twice as much water?
Cold Balloons • Submerge an inflated balloon in liquid nitrogen (-196°C) MIT_Physics_Demo_--_Balloons_in_Liquid_Nitrogen.avi • What is held constant? • Pressure? • Volume? • Temperature? • Amount of gas? • What is the relationship between temperature and volume? • As the temperature decreases, volume decreases • Direct Relationship
Two Equations to Rule Them All • P = (constant) T at a constant V and n • P = (constant) n at a constant V and T • P = (constant) / V at a constant n and T
Ideal Gas Law • R = Universal Gas Constant
Combined Gas Law • If we have a sealed container • The number of moles is constant
Practice Problems • What is the pressure in kilopascals inside a 20.0L container filled with 8.92mol of hydrogen gas at 25°C?
Practice Problems • A 50.0L cylinder of argon gas at 19.8 atm has a temperature of 19.0°C. What volume of air could it displace in a room at 0.974atm pressure at the same temperature?
Practice Problems • A 550mL balloon is placed in a Bell jar attached to a vacuum pump. The vacuum pump is accidentally reversed so that the pressure increases inside the Bell jar from 0.987atm to 3.50atm before the Bell jar explodes. What is the volume of the balloon in the Bell jar before the jar explodes?
Practice Problems • A certain car tire has a volume of 29.0L. The maximum rated operating pressure is 80.0psi. The temperature of a hot tire can reach as high as 105°C on a race track. What is the maximum amount of gas that should be pumped into the cold tire to ensure it doesn’t burst in the middle of a race? (1atm = 14.7 lbs/in2)
Practice Problems • What mass of nitrogen is in a cylinder with a volume of 3.25L and a pressure of 15500 mmHg at 25°C?
Methanol/air fires can burn at temperatures as high as 1900°C. After shooting an 18.9L methanol rocket down the hall, it is quickly stoppered to seal the remaining gas in the container. What is the pressure inside the container once it cools back down to room temperature? Assume that atmospheric pressure is 1.00atm.
The typical pressure in Boone is around 670 torr. Opening shampoo after driving from Winston-Salem (where the typical pressure is around 740torr) to Boone is occasionally a slightly hazardous experience. If you have a bottle with a 150mL of head space in Winston-Salem, what volume should it occupy in Boone?
Methanol/air fires can burn at temperatures as high as 1900°C. After shooting an 18.9L methanol rocket down the hall, it is quickly stoppered to seal the remaining gas in the container. What is the pressure inside the container once it cools back down to room temperature (25.0°C)? Assume that atmospheric pressure is 1.00atm. • What mass of nitrogen is in a cylinder with a volume of 3.25L and a pressure of 15500 mmHg at 25.0°C? • The typical pressure in Boone is around 670 torr. Opening shampoo after driving from Winston-Salem (where the typical pressure is around 740torr) to Boone is occasionally a slightly hazardous experience. If you have a bottle with a 150mL of head space in Winston-Salem, what volume should it occupy in Boone? • A certain car tire has a volume of 29.0L. The maximum rated operating pressure is 80.0psi. The temperature of a hot tire can reach as high as 105°C on a race track. What is the maximum amount of gas that should be pumped into the cold tire to ensure it doesn’t burst in the middle of a race? (1atm = 14.7 lbs/in2)
Mixtures of Gasses • How do we deal with them? • The total pressure is the sum of the pressure of each component. • Pressures are additive • Ptotal = P1 + P2 + P3 + P4 + … • P1 = Pressure of Component 1 = Partial Pressure of 1 • Dalton’s Law of Partial Pressures
Practice • The atmosphere is made up of many gasses. The three largest components are nitrogen, oxygen, and argon gasses with partial pressures of 0.78084atm, 0.20946atm, and 0.00934atm respectively. What is the partial pressure of all of the minor components of air combined?
Collecting a Gas Over Water • Vapor Pressure of Water – the partial pressure of water in an air sample that is saturated with water vapor. • Temperature Dependent
Practice • How many moles of oxygen gas were produced by the decomposition of potassium chlorate with a manganese(IV) oxide catalyst if 250mL of oxygen was collected over water at 740mmHg and 22.0°C? The vapor pressure of water at 22.0°C is 19.83mmHg. How many grams of potassium chlorate were decomposed?
Kinetic Molecular Theory for Ideal Gasses • A rather simple but powerful system • Can derive the ideal gas law from it • Describes gases pretty well under certain conditions • Not perfect and has limitations
Kinetic Molecular Theory for Ideal Gases • Gases consist of large numbers of particles (atoms or molecules) that are in continuous random motion. • The combined volume of all the molecules of the gas is negligible compared to the total volume in which the gas is contained. • Attractive and repulsive forces between gas molecules are negligible. • Energy is transferred between particles during collisions. These collisions are completely elastic (there is no loss in kinetic energy.) • The average kinetic energy of the particles is proportional to the absolute temperature. At a given temperature, all different gases have the same average kinetic energy.