Water
Water. --a special liquid for a special set of students. Physical Parameters of Water. Formula FM Shape Polar? Density . Physical Parameters of Water. Formula H 2 O FM 18.02 g/mol Shape bent, 104.5 o Polar? Yes Density 1.00 g/ml. Thermal characteristics of Water. MP

Water
E N D
Presentation Transcript
Water --a special liquid for a special set of students
Physical Parameters of Water • Formula • FM • Shape • Polar? • Density
Physical Parameters of Water • Formula H2O • FM 18.02 g/mol • Shape bent, 104.5o • Polar? Yes • Density 1.00 g/ml
Thermal characteristics of Water • MP • BP • C • Hfus • Hvap
Thermal characteristics of Water • MP 0.0oC • BP 100.0oC • C 4.18 J/goC • Hfus6 kJ/mol, 334 J/g • Hvap41 kJ/mol, 2300 J/g
Is this special? • Consider Table 17.1—MP and BP of molecules of similar size (and HF) • Formula FM(g/mol) MP (oC) BP (oC) • CH4 • NH3 • H2O • HF • Ne
Is this special? • Consider Table 17.1—MP and BP of molecules of similar size (and HF): p 480 • Formula FM(g/mol) MP (oC) BP (oC) • CH4 16 • NH3 17 • H2O 18 = 18 g/mol ±11% • HF 20 • Ne 20
Is this special? • Consider Table 17.1—MP and BP of molecules of similar size (and HF): p 480 • Formula FM(g/mol) MP (oC) BP (oC) • CH4 16 -183 • NH3 17 -78 • H2O 18 0 • HF 20 -83 • Ne 20 -249
For the covalent hydrogen compounds of the second period: Water!
Is this special? • Consider Table 17.1—MP and BP of molecules of similar size (and HF): p 480 • Formula FM(g/mol) MP (oC) BP (oC) • CH4 16 -183 -164 • NH3 17 -78 -33 • H2O 18 0 100 • HF 20 -83 20 • Ne 20 -249 -246
For the covalent hydrogen compounds of the second period: Water!
Is this special? • Consider Table 17.1—MP and BP of molecules of similar size (and HF): p 480 • Formula FM(g/mol) Liquid Range • CH4 16 19 • NH3 17 45 • H2O 18 100 • HF 20 103 • Ne 20 3
For the covalent hydrogen compounds of the second period: Water!
Consider KMT: • Melting occurs when particles have enough motion to escape their solid structure
Consider KMT: • Melting occurs when particles have enough motion to escape their solid structure • A substance whose particles cling together better has a higher melting point
Consider KMT: • Boiling occurs when particles have enough motion to escape their liquid neighbors
Consider KMT: • Boiling occurs when particles have enough motion to escape their liquid neighbors • A substance whose particles cling together better has a higher boiling point
Consider KMT: • The liquid range is all of those temperatures where the particles move around each other, but are unlikely to escape
Consider KMT: • The liquid range is all of those temperatures where the particles move around each other, but are unlikely to escape • A substance whose particles cling together better, even while moving, has a larger liquid range.
Therefore… • Water molecules stick together very well—in a solid, and as a liquid.
Why do molecules stick together? • Attractions between molecules are called intermolecular forces (IM forces) • Different types of substances have different types of IM forces • Some forces are stronger than others.
Non-polar molecules… Showdispersion forces • very weak • very brief, small charge imbalances due to the motion of electrons. • They unbalance and attract their neighbors.
Polar molecules… Show dipole interactions • fairly weak. • permanent, small charge imbalances due to the polarity of their bonds. • They attract their polar neighbors.
(But, not all polar bonds are created equal) • When hydrogen is the less electronegative end of a polar bond: d+ d- H Cl --the hydrogen is more positive --it is losing custody of its last electron
Polar molecules with hydrogen… Show hydrogen bonding • strongest of the weak bonds. • permanent, larger charge imbalances than other polar bonds. • They attract their polar neighbors better.
Strong IM forces include… Ionic bonds (in ionic compounds) Metallic bonds (in pure metals and alloys) Covalent bonds (in covalent network solids) (None of these particles are molecules, but they are still called intermolecular forces.)
Why do particles stick together? • In order, from weakest to strongest: Dispersion Forces Dipole Interactions Hydrogen Bonding Ionic Bonds Metallic Bonds Covalent Bonds
Why do particles stick together? • In order, from weakest to strongest: Dispersion Forces —between non-polar molecules Dipole Interactions —between polar molecules Hydrogen Bonding -between polar molecules w/H Ionic Bonds —between ions Metallic Bonds —between metal atoms Covalent Bonds —in a network solid
Why do particles stick together? If you are given a substance: • --describe the type of substance • --describe the strongest IM force between the particles • --you may be asked to compare it to another substance
What kind of substance? • barium • chlorine • tin (II) chloride • sulfur dioxide • solid sulfur • helium • nitrogen dioxide • iron • sodium oxide • iodine • barium sulfide • sulfuric acid
Why do particles stick together? • Watch out for a trick question.
Why do particles stick together? • Watch out for a trick question. --Ready?
Quiz Q: What holds water together?
Quiz Q: What holds water together? A: HA! It’s a trick question! There are TWO answers, both important.
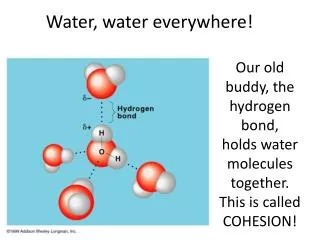
Answer 1: • Polar covalent bonds between the hydrogen and oxygen atoms hold the atoms together as water molecules !
! • Answer 2: • Hydrogen bonding, due to the hydrogen being the less electronegative atom of a polar covalent bond, attracts a water molecule to its neighbors as a liquid or a solid. (Please notice that the first answer leads to the second.)
Is this special? • Consider Table 17.1—MP and BP of molecules of similar size (and HF) • Formula Type of substance • CH4 • NH3 • H2O • HF • Ne
Is this special? • Consider Table 17.1—MP and BP of molecules of similar size (and HF) • Formula Type of substance • CH4 non-polar covalent molecule • NH3 polar covalent molecule • H2O polar covalent molecule • HF polar covalent molecule • Ne non-polar individual atoms
Is this special? • Consider Table 17.1—MP and BP of molecules of similar size (and HF) • Formula Type of IM Forces • CH4 dispersion forces • NH3 hydrogen bonding • H2O hydrogen bonding • HF hydrogen bonding • Ne dispersion forces
Which has stronger intermolecular forces, NaCl or HCl? • NaCl: ionic compound (Na+ and Cl- ions) HCl: polar covalent molecule (linear, with H in a polar bond).
Which has stronger intermolecular forces, NaCl or HCl? • NaCl: ionic compound (Na+ and Cl- ions) HCl: polar covalent molecule (linear, with H in a polar bond). • NaCl: held together by ionic bonds HCl molecules: attracted to each other by hydrogen bonds.
Which has stronger intermolecular forces, NaCl or HCl? • NaCl: ionic compound (Na+ and Cl- ions) HCl: polar covalent molecule (linear, with H in a polar bond). • NaCl: held together by ionic bonds HCl molecules: attracted to each other by hydrogen bonds.. • The ionic bonds in NaCl are stronger than hydrogen bonds between HCl molecules
What kind of IM forces? • barium • chlorine • tin (II) chloride • sulfur dioxide • solid sulfur • helium • nitrogen dioxide • iron • sodium oxide • iodine • barium sulfide • sulfuric acid
Rank in order of strength of IM forces. • barium • chlorine • tin (II) chloride • sulfur dioxide • solid sulfur • helium • nitrogen dioxide • iron • sodium oxide • iodine • barium sulfide • sulfuric acid
Compare N2 and CO • What type of substance? • What type of IM forces • Which is stronger? • What will this do to the MP and BP?
There is an overlap. • The strongest dispersion forces are stronger than average dipole interactions • In general, a larger molecule has stronger dispersion forces. • There is a big overlap between ionic and metallic bonds.