The mole
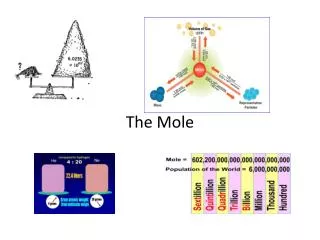
The mole. What is a Mole. Chemists use the mole to count microscopic particles. How many socks come in a pair? 2 How many eggs are in a dozen? 12 How many eggs come in a gross? 144 How many pencils come in a ream? 500. So how many atoms come in a mole?. 602,213,670,000,000,000,000,000.

The mole
E N D
Presentation Transcript
What is a Mole • Chemists use the mole to count microscopic particles. • How many socks come in a pair? • 2 • How many eggs are in a dozen? • 12 • How many eggs come in a gross? • 144 • How many pencils come in a ream? • 500
So how many atoms come in a mole? • 602,213,670,000,000,000,000,000. • Seriously • This number was created by an Italian physicist and lawyer named Amedeo Avogadro. • Its called Avogadro’s Number. • Is there an easier way to write it? • 6.02 x 1023 items = 1 mole
Converting between moles and number of particles • Think about eggs • 1 dozen eggs = 12 eggs • Conversion factor: • 12 eggs/1 dozen eggs • OR • 1 dozen eggs/12 eggs • So if you have 3.5 dozen eggs how many eggs do you have? • 3.5 dozen x (12 eggs)/1 dozen = 42 eggs
If you have 3.5 moles of sugar how many particles of sugar do you have? • 1 mole of sugar = 6.02 x 1023 particles of sugar. • Set up the problem: • 3.5 moles x (6.02 x 1023 particles/1 mole) • Solve: • 2.107 x 1024
Zinc (Zn) is used to form a corrosion-inhibiting surface on galvanized steel. Determine the number of Zn atoms in 2.50 moles of Zn. • 1.51 x 1024 atoms • Calculate the number of molecules in 11.5 mol of water. • 6.92 x 1024 molecules • Silver nitrate Ag(NO3)2 is used to make several different compounds used in photographic films. How many molecules of silver nitrate are there in 3.25 moles of Ag(NO3)2? • 1.96 x 1024 • How many atoms of oxygen are there in 5.0 mol of oxygen gas? • 6.02 x 1024 atoms
Converting from number of particles to moles • Calculate the number of moles of Zinc that contains 4.50 x 1024 atoms. • 0.914 moles • How many moles can be made up from 5.57 x 1024 atoms of Al? • 9.55 moles • How many moles can be made up from 2.5 x 1025 atoms of Fe? • 41.5 moles
What would have more mass a dozen eggs or a dozen elephants? • Why would a dozen elephants have more mass? • Just like elephants and eggs certain atoms are bigger than others. • For example Neon is much bigger than Helium. • Which would have more mass 1 mole of Neon or 1 mole of Helium?
Molar Mass • The molar mass of an element is the mass in grams of one mole of that element. • Why is this important? • Molar mass can be used to calculate the number of atoms with out using a microscope.
Using molar mass • If one dozen jelly beans has a mass of 35 g how much mass does 5 dozen jelly beans have? • 175 g • To convert from Moles to mass you multiply by the molar mass. • What is the mass of 3.oo moles of copper? • 191 g cu. • To convert from mass to moles you divide by the molar mass.
Example Problems • Chromium (Cr), a transition element, is a component of chrome plating. Chrome plating is used on metals and steel alloys to control corrosion. Calculate the mass in grams of 0.045 moles of Cr. • 2.34 g Cr
Calcium (Ca), the fifth most abundant element on earth, is always found combined with other atoms because of its high reactivity. How many moles of calcium are in 525 g Ca? • 13.1 moles Ca
Converting between mass and atoms • The next step is converting a given mass of an element into a number of atoms. • If we have 550 g of jelly beans and there are 35 g of jelly beans in a dozen how many dozen jelly beans do we have? • 16 dozen • How many jelly beans are there in 16 dozen? • 192
Mass-to-Atom Conversion • Gold (Au) is one of a group of metals called the coinage metals. How many atoms of gold are there in a U.S. gold eagle coin with a mass of 31.1 g? • 9.51 x 1022
Atom-to-Mass Conversion • Helium (He) is an unreactive noble gas found underground. A party balloon contains 5.5 x 1022 atoms of helium gas. What is the mass, in grams, of the helium? • 0.366 g
Homework • Textbook (sorry) • P. 328 # 15(a&b), 16(a&b) • P. 329 # 17(a&b), 18(a&b) • P. 331 # 19(a&b), 20(a&b)
Chemical Formulas and the Mole • In the compound CCl2F2 how many atoms do we have? • C: 1 • Cl: 2 • F: 2 • So if we have one mole of CCl2F2 we have one mole of carbon, two moles of Chlorine and one mole of Fluorine.
Molar Mass of a Compound • Determine the molar mass of each of the following compounds: • NaOH • CaCl2 • KC2H3O2 • C2H5OH • HCN • CCl4 • (NH4)3PO4
Converting moles to mass • The characteristic odor of garlic is due to allyl sulfide (C3H5)2S. What is the mass of 2.5 moles of allyl sulfide? • What is the mass of 3.25 mol of H2SO4?
Converting Mass to Moles • Calculate the number of moles in 325 g of Calcium hydroxide Ca(OH)2? • Calculate the number of moles in 22.6 g of AgNO3.
Mass to Number of Particles • Aluminum chloride (AlCl3) is used in refining petroleum and manufacturing rubber. How many Aluminum Chloride molecules are present in 35.6 g of AlCl3 • How many Al atoms are there? • How many Cl atoms are there?
Example • Aluminum oxide (Al2O3) when dissolved in water breaks apart into ions (Al3+ and O2-) how many moles of Al3+ will be produced when 1.5 moles of Al2O3 are dissolved in water?
Examples • What is the molar mass of ethanol C2H5OH? • How many ethanol molecules are present in 45.6 g? • How many carbon atoms are there in 45.6 g of ethanol? • How many hydrogen atoms are there in 45.6 g of ethanol? • How many oxygen atoms are there in 45.6 g of ethanol? • A sample of sodium sulfite Na2SO3 has a mass of 2.25 g • How many molecules of sodium sulfite are there? • How many atoms of sodium are there? • How many atoms of Sulfur are there? • How many atoms of oxygen are there?
Percent Composition • A compound is made up of one or more atoms bonded together. • Each of the atoms contributes mass to the compound. • Example: • The molar mass of NaOH is • Na – 22.98 g/mol • O – 16 g/mol • H – 1 g/mol • Total: 39.98 g/mol
Calculating Percent Composition • Percent composition is calculated by dividing the mass of an individual element by the mass of the whole compound and then multiplying by 100. • NaOH: • Percent Composition of Na: • (22.98/39.98) x 100 = 57.5% • Percent Composition of O: • (16/39.98) x 100 = 40% • Percent Composition of H: • (1/39.98) x 100 = 2.5%
Compounds with multiple atoms of the same element • NaHCO3 • Molar Mass: 84.01 g/mol • Percent Composition of Na: • (22.98/84.01) x 100 = 27.37% • Percent Composition of H: • (1/84.o1) x 100 = 1.2% • Percent Composition of C: • (12.01/84.01) x 100 = 14.3% • Percent Composition of O: • (48/84.01) x 100 = 57.14%
Empirical Formula • When we know a compounds percent composition we can determine it’s formula. • The empirical formula of a compound is the formula with the smallest whole-number ratio of elements. • Find the empirical formula for a compound that is 40.05% S and 59.95% O. • If we assume that we have 100 g of this compound: • How many grams of Sulfur do we have? • 40.05 g • How many grams of Oxygen do we have? • 59.95 g • Convert from grams to moles: • 40.05 g S x 1 mol S/32.07 g S = 1.249 mol • 59.95 g O x 1 mol O/16.00 g O = 3.747 mol
S1.249O3.747 • SO3 • Sometimes the empirical formula is not the actual formula for the compound. • Find the empirical formula for hydrogen peroxide which has a molar mass of 34 g/mol and is 5.9% Hydrogen and 94.1% Oxygen
Molecular Formula • A compounds molecular formula is its actual formula (not always the same as empirical formula) • To determine the true molecular formula for a compound its molar mass must be known (either looked up or found experimentally). • Once we know the molar mass and the empirical formula we can find the true formula of the compound.
Example: • Acetylene is a highly flammable compound used in blow torches. Acetylene has a molar mass of 26.04 g/mol. It is 92.2% Carbon and 7.8% Hydrogen. What is the chemical formula of Acetylene? • Step 1: Find empirical formula: • CH • Step 2: Calculate molar mass of empirical formula. (If the molar mass is the same as the actual molar mass then the empirical formula is the true formula.) • 13.02 g/mol • Step 3: Divide real molar mass by the molar mass of the empirical formula. • 26.04/13.02 = 2.000 • Step 4: Multiply all atoms in the empirical formula by the answer to the true formula. • C2H2