The production procedure
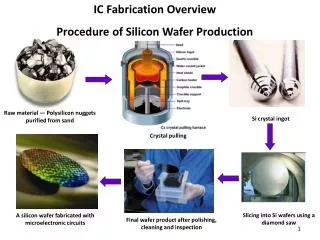
The production procedure. Step 1 Hybridoma cell production Hybridoma cell( 杂交瘤细胞 ): the cell which made by fusing a specific antibody-producing B cell with a myeloma (B cell cancer) cell that is selected for its ability to grow in tissue culture and for an absence of antibody chain synthesis.

The production procedure
E N D
Presentation Transcript
The production procedure Step 1 Hybridoma cell production Hybridoma cell(杂交瘤细胞): the cell which made by fusing a specific antibody-producing B cell with a myeloma(B cell cancer) cell that is selected for its ability to grow in tissue culture and for an absence of antibody chain synthesis.
Details of production • (1) Immunisation of a mouse(2) Isolation of B cells from the spleen(3) Cultivation of myeloma cells(4) Fusion of myeloma and B cells(5) Separation of cell lines(6) Screening of suitable cell lines(7)in vitro(a) or in vivo(b) multiplication(8) Harvesting
Monoclonal antibodies are typically made by fusing myeloma cells(骨髓瘤细胞) with the spleen cells from a mouse that has been immunized with the desired antigen. • The selective culture medium is called HAT medium because it contains hypoxanthine, aminopterin, and thymidine. This medium is selective for fused (hybridoma) cells. Unfused myeloma cells cannot grow because they lack HGPRT (an enzyme necessary for the salvage synthesis--a pathway in which nucleotides are synthesized from intermediates in the degradative pathway for nucleotides of nucleic acids), and thus cannot replicate their DNA. Unfused spleen cells cannot grow indefinitely because of their limited life span. Only fused hybrid cells, referred to as hybridomas, are able to grow indefinitely in the media because the spleen cell partner supplies HGPRT and the myeloma partner has traits that make it immortal (as it is a cancer cell).
This mixture of cells is then diluted and clones are grown from single parent cells on microtitre wells. The antibodies secreted by the different clones are then assayed for their ability to bind to the antigen (with a test such as ELISA or Antigen Microarray Assay) or immuno-dot blot. The most productive and stable clone is then selected for future use. A Dot blot (or Slot blot) is a technique in molecular biology used to detect biomolecules. Schematic of the use of two ASO probes on duplicate Dot-blot filters.
The hybridomas can be grown indefinitely in a suitable cell culture media, or they can be injected in mice (in the peritoneal cavity(腹膜腔), the gut), they produce tumors containing an antibody-rich fluid called ascites fluid(腹水). The medium must be enriched during selection to further favour hybridoma growth. This can be achieved by the use of a layer of feeder fibrocyte cells or supplement medium such as briclone (a kind of medium for use in the stages following fusion in hybridoma production. ). Production in cell culture is usually preferred as the ascites technique is painful to the animal and if replacement techniques exist, this method is considered unethical.
Step2 Purification of monoclonal antibodies • WHY? The contaminants in the cell culture sample would consist primarily of media components such as growth factors, hormones, and transferrins(铁传递蛋白). In contrast, the in vivo sample is likely to have host antibodies, proteases, nucleases, nucleic acids, and viruses. In both cases, other secretions by the hybridomas such as cytokines may be present. There may also be bacterial contamination and, as a result, endotoxins which are secreted by the bacteria.
HOW? • To achieve maximum purity in a single step, affinity purification can be performed, using the antigen to provide exquisite specificity for the antibody. In this method, the antigen used to generate the antibody is covalently attached to an agarose support. If the antigen is a peptide, it is commonly synthesized with a terminal cysteine which allows selective attachment to a carrier protein, such as KLH during development and to the support for purification. The antibody-containing media is then incubated with the immobilized antigen, either in batch or as the antibody is passed through a column, where it selectively binds and can be retained while impurities are washed away. An elution with a low pH buffer or a more gentle, high salt elution buffer is then used to recover purified antibody from the support. • To further select for antibodies, the antibodies can be precipitated out using sodium sulfate or ammonium sulfate. Antibodies precipitate at low concentrations of the salt, while most other proteins precipitate at higher concentrations. The appropriate level of salt is added in order to achieve the best separation. Excess salt must then be removed by a desalting method such as dialysis. • The final purity can be analyzed using a chromatogram. Any impurities will produce peaks, and the volume under the peak indicates the amount of the impurity. Alternatively, gel electrophoresis and capillary electrophoresis can be carried out. Impurities will produce bands of varying intensity, depending on how much of the impurity is present.
To simplify: • Affinity purification • Antibody precipitation • Using a chromatogram to identify the impurities
Step3 Recombinant • The production of recombinant monoclonal antibodies involves technologies, referred to as repertoirecloning or phage display/yeast display. Recombinant antibody engineering involves the use of viruses or yeast to create antibodies, rather than mice. These techniques rely on rapid cloning of immunoglobulin gene segments to create libraries of antibodies with slightly different amino acid sequences from which antibodies with desired specificities can be selected. These techniques can be used to enhance the specificity with which antibodies recognize antigens, their stability in various environmental conditions, their therapeutic efficacy, and their detectability in diagnostic applications. Fermentation chambers have been used to produce these antibodies on a large scale.