Global Market Trends in Alzheimer’s Disease Therapies: Opportunities and Challenges
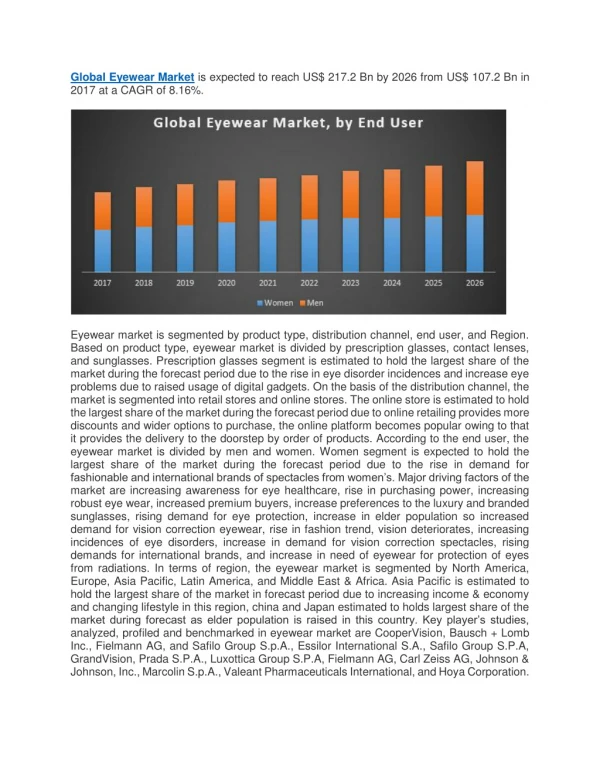
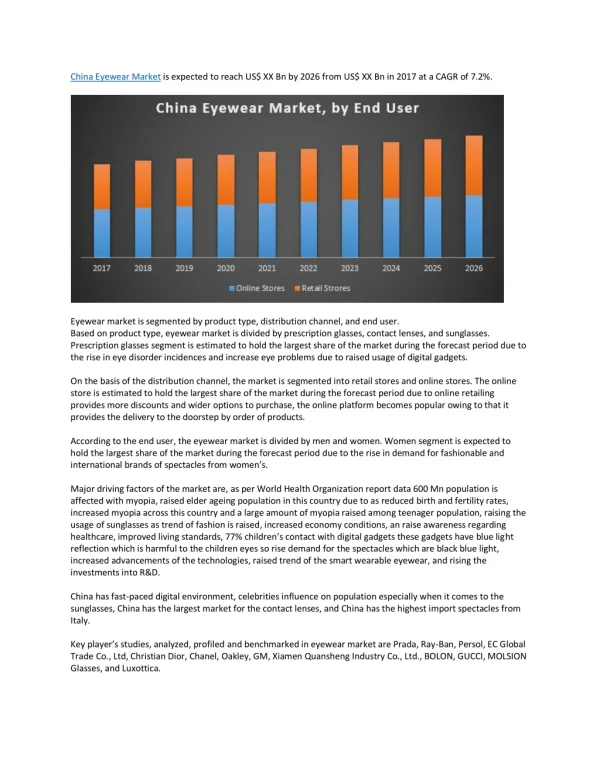
The Alzheimer’s disease therapy market is experiencing shifts due to patent expirations and the influx of generics, leading to decreased global spending despite increased treatment volumes driven by demographic changes. Aricept (Donepezil) remains the leading product globally but faces competition from generics. Sales distribution highlights North America (38%) as the largest market, followed by Europe (30%), while regions with younger populations like Latin America (4%) lag behind. The sector sees significant R&D investment, with a notable decline in spending on branded therapies first reported in 2011.

Global Market Trends in Alzheimer’s Disease Therapies: Opportunities and Challenges
E N D
Presentation Transcript
Global market facts and trends regarding Alzheimer‘s disease therapies: • Patent losses and entrance of generics in the market lead to lower global spending in this therapy area; however volume increases asa consequence of demographic change • Aricept (Donepezil) is still the leading product worldwide, however exposed to generic competition • According to regions/countries the distribution of sales shows: North America on the top (38%, one-year-period from April 2011 upto march 2012), followed by Europe (30%), Africa, Asia, Australasia (28%) and Latin America (4%). This distribution also reflects the age structures, because Latin America has a quite young population, compared internationally • The Alzheimer‘s disease area is characterized by an intensive R&D with many products under development Spending on predominantly brand-dominated Alzheimer’s disease therapies declined for the first time in 2011 Alzheimer’s Therapies Global Spending and Volume, 2001-2011 VOLUME (STANDARD UNITS BN) SALES (US$BN) Source: IMS MIDAS®, Dec 2011 Four brands have been losing exclusivity in key countries since 2010, opening this therapy space to generic competition Alzheimer’s Therapy Sales Exposed to Generic Competition (2010-2015) US Spain Japan Europe Canada Europe US US PRE-EXPIRY SALES* (US$BN) Europe Canada Source: IMS MIDAS®, March 2012 * Molecule sales in year prior to loss of exclusivity. INFOGRAFIK 09_2/2012