Lecture 2 Atoms and Molecules
330 likes | 608 Vues
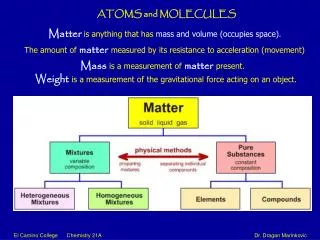
Lecture 2 Atoms and Molecules. Atoms. Matter is any substance in the universe that has mass and occupies space All matter is composed of extremely small particles called atoms. Every atom has the same basic structure Core nucleus of protons and neutrons Orbiting cloud of electrons.

Lecture 2 Atoms and Molecules
E N D
Presentation Transcript
Atoms • Matter is any substance in the universe that has mass and occupies space • All matter is composed of extremely small particles called atoms • Every atom has the same basic structure • Core nucleus of protons and neutrons • Orbiting cloud of electrons
Atoms • Atomic number • Number of protons • Atomic mass • Number of protons and neutrons • Element • A substance that cannot be broken down by ordinary chemical means
Different atomic mass Same atomic number Isotopes • Isotopes are atoms with the same number of protons but different numbers of neutrons 99% of all carbon
Radioactive Decay Radioactive isotope dating • The nucleus of an unstable isotope breaks down into particles with lower atomic numbers • Radioactive isotopes are used in • 1. Medicine • Tracers are taken up and used by the body • Emissions are detected using special lab equipment • 2. Dating fossils • The rate of decay of a radioactive element is constant • The amount of decay can be used to date fossils
Energy • The capacity to do work (put matter into motion) • Types of energy • Kinetic – energy in action • Potential – energy of position; stored (inactive) energy PLAY Energy Concepts
Electrons • Electrons have energy due to their relative orbital position (potential energy) • Electron shells, or energy levels, surround the nucleus of an atom • Valence shell – outermost energy level containing chemically active electrons • Bonds are formed using the electrons in the outermost energy level
The Octet Rule • Inert elements have their outermost energy level fully occupied by electrons • Octet rule – except for the first shell which is full with two electrons, atoms interact in a manner to have eight electrons in their valence shell • Reactive elements do not have their outermost energy level fully occupied by electrons
Molecules • Amolecule is a group of atoms held together by energy • The holding force is called a chemical bond • There are three kinds of chemical bonds • Ionic bonds • Covalent bonds • Hydrogen bonds
Ionic Bonds • Ionic bonds form between atoms by the transfer of one or more electrons • Ionic compounds form crystals instead of individual molecules • Example: NaCl (sodium chloride) • Two key properties • Strong: But not as strong as covalent bonds • Not directional: They are not formed between particular ions in the compound
Covalent Bonds • Covalent bonds are formed by the sharing of two or more electrons • Electron sharing produces molecules • Two key properties • Strong: The strength increases with the number of shared electrons • Very directional: They are formed between two specific atoms Water molecules contain two covalent bonds
Comparison of Bonds • Electrons shared equally between atoms produce nonpolar molecules • Electrons shared unequally produces polar molecules • Atoms with six or seven valence shell electrons are electronegative • Atoms with one or two valence shell electrons are electropositive
Hydrogen Bonds • Formed by the attraction of opposite partial electric charges between two polar molecules • Too weak to bind atoms together • Common in dipoles such as water • Responsible for surface tension in water • Important as intramolecular bonds, giving the molecule a three-dimensional shape Hydrogen bonding in water molecules
Chemical Reactions • Occur when chemical bonds are formed, rearranged, or broken • Are written in symbolic form using chemical equations • Chemical equations contain: • Number and type of reacting substances, and products produced • Relative amounts of reactants and products
Patterns of Chemical Reactions • Combination reactions:Synthesis reactions which always involve bond formation A + B AB • Decomposition reactions: Molecules are broken down into smaller molecules AB A + B • Exchange reactions: Bonds are both made and broken AB + C AC + B • All chemical reactions are theoretically reversible A + B AB AB A + B • If neither a forward nor reverse reaction is dominant, chemical equilibrium is reached
Oxidation-Reduction (Redox) Reactions • Reactants losing electrons are electron donors and are oxidized • Reactants taking up electrons are electron acceptors and become reduced • Generally, the atom that is reduced contains the most energy
Energy Flow in Chemical Reactions • Exergonic reactions – reactions that release energy • Endergonic reactions – reactions whose products contain more potential energy than did its reactants
Factors Influencing Rate of Chemical Reactions • Temperature – chemical reactions proceed quicker at higher temperatures • Particle size – the smaller the particle the faster the chemical reaction • Concentration – higher reacting particle concentrations produce faster reactions • Catalysts – increase the rate of a reaction without being chemically changed • Enzymes – biological catalysts
Water molecules are polar molecules They can thus form hydrogen bonds with each other and with other polar molecules Each hydrogen bond is very weak However, the cumulative effect of enormous numbers can make them quite strong Hydrogen bonding is responsible for many of the physical properties of water Heat Storage A large input of thermal energy is required to disrupt the organization of liquid water This minimizes temperature changes Ice Formation At low temperatures, hydrogen bonds don’t break Water forms a regular crystal structure that floats High Heat of Vaporization At high temperatures, hydrogen bonds do break Water is changed into vapor Hydrogen Bonds Give Water Unique Properties PLAY Water Transport
Cohesion Attraction of water molecules to other water molecules Example: Surface tension Adhesion Attraction of water molecules to other polar molecules Example: Capillary action Water strider Hydrogen Bonds Give Water Unique Properties
High Polarity Polar molecules are termed hydrophilic Water-loving All polar molecules that dissolve in water are termed soluble Nonpolar molecules are termed hydrophobic Water-fearing These do not form hydrogen bonds and are therefore not water soluble Hydrogen Bonds Give Water Unique Properties
+ H2O OH– H+ hydroxide ion hydrogen ion Water Ionizes • Covalent bonds within a water molecule sometimes break spontaneously • This process of spontaneous ion formation is called ionization • It is not common because of the strength of covalent bonds
_ pH log [H+] = pH • A convenient way to express the hydrogen ion concentration of a solution • The pH scale is logarithmic • A difference of one unit represents a ten-fold change in H+ concentration • Acid: Dissociates in water to increase H+concentration • Base: Combines with H+when dissolved inwater
Buffers • Hydrogen ion reservoirs that take up or release H+ as needed • The key buffer in blood is an acid-base pair