Baker-Venkataraman Rearrangement
290 likes | 1.87k Vues
Baker-Venkataraman Rearrangement. Eiman Wael Jawdat Imran . Table of Contents. Objective. The presentation will give information about the (Baker-Venkataraman rearrangement) as reaction, mechanism, application and recent literature . Background.

Baker-Venkataraman Rearrangement
E N D
Presentation Transcript
Baker-Venkataraman Rearrangement Eiman Wael Jawdat Imran
Objective The presentation will give information about the (Baker-Venkataraman rearrangement) as reaction, mechanism, application and recent literature
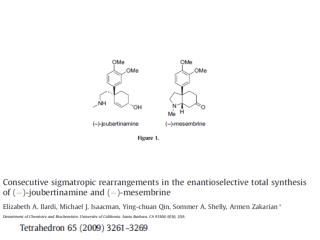
Background The base-catalyzed rearrangement of aromatic ortho-acyloxyketones to the corresponding aromatic ß-diketones is known as the Baker-Venakataraman rearrangement. ß-Diketones are important synthetic intermediates, and they are widely used for synthesis of chromones, flavones, isoflavones, and coumarins. The most commonly used bases are the following: KOH, potassium tert-butoxide in DMSO. Na metal in toluene, soduim or potassium hydride, pyridine, and triphenylmethylsodium. 1
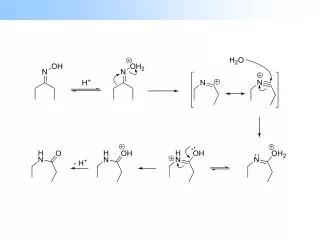
Reaction and Mechanism Reaction the Baker-venkataraman rearrangement is the chemical reaction of 2-acetoxyacetophenones with base to form 1,3-diketones. Mechanism With a base being a catalyst, it attacks hydrogen atom in Acetophenone and enolate is formed. then, the enolate group attacks the carbon in the phenol ester to form a cyclic alkoxide, Finally, it opens up a phenolate which is reprocessed by acid to undergo protonation. 2
Application and Recent Literature Recent Literature Directed ortho metalation-cross coupling links. Carbamoyl rendition of the baker-venkataraman rearrangement. Regiospecific route to substituted 4-hydroxocyoumarins. 3
Application The Baker-Venkataraman rearrangement is often used to synthesize chromones and flavones. 2 In the laboratory of K. Krohn, the total synthesis of aklanonlc acid and its derivatives was undertaken, utilizing the Baker-venkataraman rearrangement of ortho-acetyl anthraquinone esters in the presence of lithium hydride. Using this method, it was possible to introduce ketide side-chains on anthraquinones in a faclle manner. 1
conclusion At the end of the research wish to be useful informaton about the interaction (Baker-Venkataraman Rearrangement). Thank you
References (1) http://books.google.com/books?id=mjpJmiZ9OZ8C&pg=PA30&dq=Baker%E2%80%93Venkataraman+rearrangement&hl=en&ei=9ACqTbPMGZSAhAfA7NDPCQ&sa=X&oi=book_result&ct=book-thumbnail&resnum=1&ved=0CCoQ6wEwAA#v=onepage&q=Baker%E2%80%93Venkataraman%20rearrangement&f=false (2) http://en.wikipedia.org/wiki/Baker%E2%80%93Venkataraman_rearrangement
(3) • A. V. Kalinin, A. J. M. Da Silva, C. C. Lopes, R. S. C. Lopes, V. Snieckus, Tetrahedron. Lett., 1998, 39, 4995-4998. • http://www.organic-chemistry.org/namedreactions/baker-venkataraman-rearrangement.shtm